User Tools
Sidebar
textbook:nrctextbook:chapter9
This is an old revision of the document!
Table of Contents
9. Gamma detectors and spectrometry
Chapter 9 from BASICS OF NUCLEAR PHYSICS AND OF RADIATION DETECTION AND MEASUREMENT - An open-access textbook for nuclear and radiochemistry students by Jukka Lehto
For the detection and measurement of gamma radiation basically two types of detectors are used: semiconductors and solid scintillators (also called phosphors). The primary physical process in both detector types is ionization of the detector material. Here, first the two detector types and then energy calibration and efficiency calibration, and finally the formation of various peaks in gamma spectra and subtraction of background are described.
9.1. Solid scintillators
In solid scintillators the detection process is based on the same principle as in liquid scintillation counting (LSC), described in a later chapter (12): radiation excites the detector material atoms (molecules in case of LSC) to a higher energy state and as the excitation state is relaxed visible light is emitted and further the light pulses are transformed into electric pulses with the aid of a photomultiplier tube. Liquid scintillators are not applicable for gamma radiation due to their low density and thus low gamma radiation stopping power and instead solid materials with higher density are used. Scintillation process was used for the radiation detection and measurement already in the beginning of the 20th century as ZnS was used to detect and count alpha radiation. There are a number of solid scintillation detector materials of which NaI is the most extensively used and discussed here in more detail. Some other materials and their benefits over NaI are discussed at the end of the section.
NaI is an effective material for gamma ray measurement since it can be manufactured in large crystals that can absorb readily penetrating gamma rays. The larger the crystal the higher is the counting efficiency. NaI as such is, however, not capable of forming light. It needs an activation by adding Tl+ ions into the crystal framework and therefore the crystal material is denoted as NaI(Tl). Tallium ions act as luminescent centers in the NaI crystal. Typically 0.001 mol-% of tallium is added to NaI. The light formation process in NaI(Tl) takes place in the following way:
- gamma radiation primarily results in the formation of electrons (e-) and holes (h+) in ionization of the detector atoms: $\gamma \rightarrow e^- + h^+$
- the electrons interact with tallium ions to form tallium atoms $e^- + \text{Tl}^+ \rightarrow \text{Tl}^0$ while the holes interact with tallium ions to form divalent tallium ions $h^+ + \text{Tl}^+ \rightarrow \text{Tl}^{2+}$
- then tallium atoms interact with holes to form excited tallium ions $h^+ + \text{Tl}^0 \rightarrow (\text{Tl}^+)^*$ and divalent tallium ions interact with electrons also forming excited tallium ions $e^- + \text{Tl}^{2+} \rightarrow (\text{Tl}^+)^*$
- finally the excitation state of tallium is relaxed and the excitation energy is emitted as visible light $(\text{Tl}^+)^* \rightarrow \text{Tl}^+ + h\nu \, (335, 420 \, \text{nm})$
Since the excitation energy level of (Tl+)* is lower than that of NaI the crystal does not absorb the formed light photons.
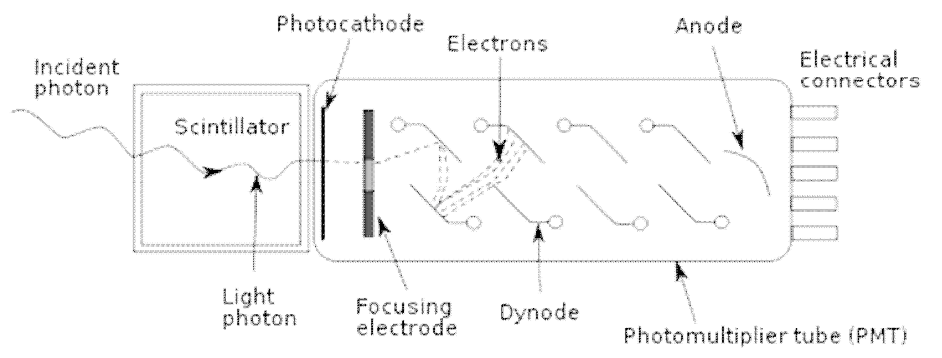
The light photons, more than 10000 for each MeV energy absorbed in the NaI(Tl) crystal, are transformed into electric pulses with a photomultiplier tube (PMT) (Figure IX.1). The number of light photons is directly proportional to the energy of gamma rays absorbed in the crystal. The light photons first hit the photocathode at the PMT end facing the NaI(Tl) crystal. Photocathode material is typically made of Cs3Sb, which releases electrons when light photons hit it. The number of released electrons is directly proportional to the number of photons hitting the photocathode. PMT multiplies the number of electrons to a countable electric pulse with the aid of successive dynodes, also made of Cs3Sb, the number of which is typically 10-14. Electric voltage is applied between each pair of dynodes which results in the increase of electron energies between the dynodes increasing release on electrons from dynodes. The high voltage through the whole PMT is 1000-2000V and the multiplication factor of electrons across the PMT is about 106. This multiplication factor is the same for all events and thus the initial number of electrons released from the photocathode is always multiplied in the PMT by the same factor. Thus, the energy information of a gamma ray absorbed in the crystal remains in all steps: formation of electrons as the primary process, formation of light photons in the crystal, formation on electrons in the photocathode and multiplication of electrons in PMT. Thus the height of the electric pulse is directly proportional to the energy of the detected gamma ray. This is, however, an ideal picture and the response varies from event to another and, instead of lines, broader peaks in the spectra are observed. The maximum of each peak, however, represents the energy of a detected gamma ray.
Figure IX.1. Photomultiplier tube (PMT) attached to a solid scintillation detector (http://chemwiki.ucdavis.edu/Analytical_Chemistry/Instrumental_Analysis/Spectrometer/Detectors/Detectors).
Since NaI(Tl) can be produced as large crystals they have good gamma ray detection efficiency, much better than what is obtained with semiconductor detectors. They can also be produced as well-type crystals in which a cylindrical whole is drilled in the middle of crystal. The sample to be counted is placed in the hole, which considerably increases counting efficiency compared to planar crystals. The drawback of solid scintillators in comparison with semiconductor detectors is their poor energy resolution. The energy resolution of NaI(Tl) detector is approximately 50-100 keV for gamma rays of energy between 2 - 0.5 MeV while with semiconductor detectors the resolution is about 50-times better (Figure IX.2). Therefore, due to overlapping peaks solid scintillators cannot be used for identification of radionuclides from samples having a large number of radionuclides. Solid scintillators are used in gamma spectrometry only when high detection efficiency is needed and when the sample does not have a large number of radionuclides. More usually, solid scintillators are used in counting of single radionuclides in a single channel mode. In this, only the pulses of the photopeak, representing the energy of the most intensive gamma transition, are counted. This is accomplished by use of voltage discriminators: the lower discriminator rejects pulses of smaller height than the set value while the upper discriminator rejects pulses of greater height than the set value.
In addition to NaI(Tl) detectors there are other types of solid scintillators, such as CsI(Tl), Bi4Ge3O12 and LaBr3(Ce). In developing solid scintillators two objectives have been sought: to improve counting efficiency and to improve energy resolution. An example of the former is Bi4Ge3O12, also called BGO, which has a better counting efficiency compared to NaI(Tl) due its higher density of 7.1 g/cm3 compared to 3.7 g/cm3 of NaI(Tl). The Ce-activated lanthanum chloride LaBr3(Ce) in turn has a much better energy resolution than NaI(Tl), 3% vs. 8% for 662 keV gamma rays. The BGO detector, however, has a lower energy resolution than NaI(Tl). Thus the choice of the detector should be done on the basis of what property is most needed, efficiency or energy resolution.
Figure IX.2. Gamma spectra collected from the same source by a semiconductor detector (left) and by a scintillation detector (right) (http://www.canberra.com/literature/fundamental-principles/).
9.2. Semiconductor detectors
Semiconductor detectors are diodes produced either of silicon or germanium, the former being used for alpha and X-ray detection and the latter for gamma detection. Germanium is more suitable to gamma detection than silicon due to its higher atomic number Z=32, and thus density, which increase the stopping power compared to silicon, the atomic number of which is only Z=14. The formation of photoelectric effect is proportional to the atomic number of the element absorbing gamma rays. Thus, for example, 0.1 MeV gamma rays are absorbed 40-times more efficiently in germanium than in silicon. The detector consists of two parts (Figure IX.3). The other part is pure Si/Ge, having four electrons in the outer shell, doped with atoms having five electrons in the outer shell, such as phosphorus. This type of semiconductor is called n-type and it acts as an electron donor. The other part, p-type, is also pure Si/Ge but now doped with atoms with three electrons in the outer shell, such as boron. This part acts as an electron acceptor with electron holes surrounding boron atoms. When these two parts are attached to each other electrons from n-type move to p-type and a narrow layer at interface, junction, becomes free of electrons and holes. This layer is called depletion layer. When now electrodes are attached to the other sides of n-type and p-type semiconductors, anode to n-type and cathode to p-type and a reverse bias voltage is applied across the system the electrons in the n-type move towards the cathode and the holes towards the anode. This results in a broadening of the depletion layer. To observe maximal depletion layer thickness very high voltages, even up to 5000 V, are used. When a gamma ray or an alpha particle hits this depletion layer it becomes conducting and an electric pulse is recorded in the external electric circuit. This pulse is amplified with a preamplifier and linear amplifier, transformed into a digital form with ADC and counted with multichannel analyzer. Since the energy to create an electron-hole pair is constant to each detector material (about 3 eV for germanium), the electric pulse is directly proportional to the energy of gamma ray or alpha particle. Thus they can be used for energy spectrometry. The semiconductor detectors act similarly to gas ionization detectors (ionization chamber and proportional counter), but the advantage of semiconductor detectors is that the formation time of an electric pulse is much shorter than in the gas ionization detectors. In addition, semiconductor detectors produce about ten times higher number of electrons (and holes) per unit energy absorbed in the detector. To be efficient for gamma ray detection the germanium detector has to be large and the depletion layer should be several centimeters wide. To observe thick depletion layer the germanium used has to be very pure, the fraction of foreign atoms being one atom per 1010 germanium atoms. These kinds of detectors are called High-purity germanium detectors (HPGe). In the early phases on germanium detector development, beginning from the 1950's, such pure germanium was not available but contained so high amounts of acceptor atoms that only a few millimeter thick depletion zones were obtainable. This naturally decreased the counting efficiency. To compensate the effect acceptor impurities Li+ ions were added to germanium crystals, called lithium-drifted germanium detectors (Ge(Li)). Lithium ions compensated the charges of acceptors and made them thus immobile. Li-drifted germanium crystals needed to be kept at liquid nitrogen temperature (-200 °C) all the time; room temperature would destroy them due to high mobility of lithium ions at higher temperatures. In the case of alpha detection the n-type facing to the source need to be very thin in order to enable penetration of alpha particles into depletion layer, which is also very thin, less than one micrometer. Alpha detectors and spectrometry are described in more detail in chapter XI.
As already mentioned, the energy resolution of germanium detector is 50-times better than that of NaI(Tl) detector and absolute resolution is about 2 keV (0.1%) for gamma rays with energies of 2 MeV, about 1.5 keV (0.15%) at 1 MeV, about 1 keV (0.2%) at 0.5 MeV and about 0.5 keV (0.5%) at 0.1 MeV. Thus germanium detectors can be used to identify gamma-emitting radionuclides from a mixture of a number of radionuclides, for example, from environmental and nuclear waste samples. Modern gamma spectrometers are provided with advanced programs, with a memory-stored library of peaks and their intensities of most gamma emitters, and thus the radionuclide identification is done automatically. Quantitative analysis of radionuclides is based on measurement of net areas of the representative peaks and using pre-determined efficiency calibration, the latter being described later in this chapter.
Figure IX.3. Structure and function of a semiconductor detector.
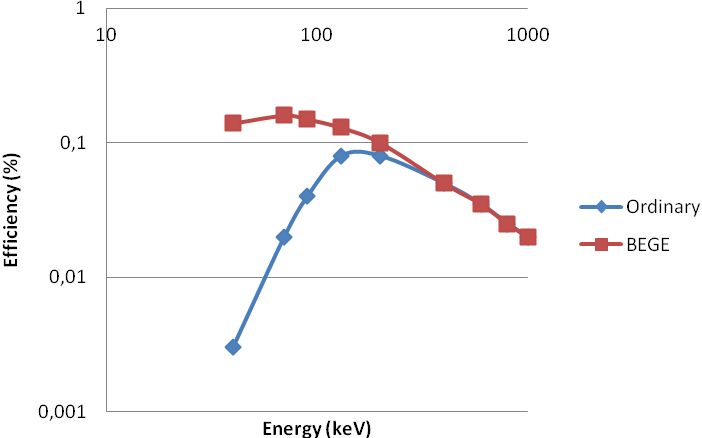
The detection efficiency of germanium detectors is dependent on the size of the detector: the larger the detector the higher the efficiency. Efficiency depends also on the gamma energy (Figure IX.4). At higher gamma energies the efficiency decreases due to penetration of gamma rays without interactions with the detector. At energies higher than about 150 keV the efficiency decreases more or less linearly when both energy and efficiency are presented on logarithmic scales. Ordinary germanium detectors are covered with an aluminum shield, which effectively absorbs low energy gamma rays. This can be seen in Figure IX.4 as a dramatic drop in efficiency of gamma ray energies below 100 keV. To overcome this and to enable also measurement of low energy gamma rays broad energy (BEGe) and low energy (LEGe) germanium detectors have been developed. These have, instead of aluminum, very thin window, made of either beryllium or carbon composite, between the source and the detector. This allows efficient detection of low energy gamma emitters, such as 210Pb (46.5 keV) and 241Am (59.5 keV) supposing their activities are high enough. Energies down to 3 keV can be measured with BEGe/LEGe detectors and even below 1 keV with ultra-low energy detectors.
Figure IX.4. Efficiencies of an aluminum-covered ordinary and broad energy (BEGE) germanium detectors with same relative efficiency as a function of gamma ray energy.
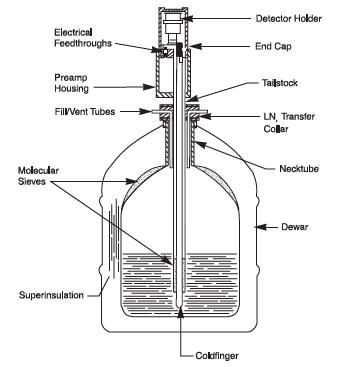
When used the germanium detectors need to be cooled to about -200°C with liquid nitrogen cryostat (Fig. IX.5) or electrically to reduce electric noice which would considerably increase the background. Modern high purity germanium detectors (HPGE) can be let to warm when not in use but the earlier generation Li-drifted germanium detectors would destroy when letting them to warm up.
Figure IX.5. Liquid nitrogen cryostat for cooling germanium detectors (http://www.canberra.com/products/detectors/germanium-detectors.asp).
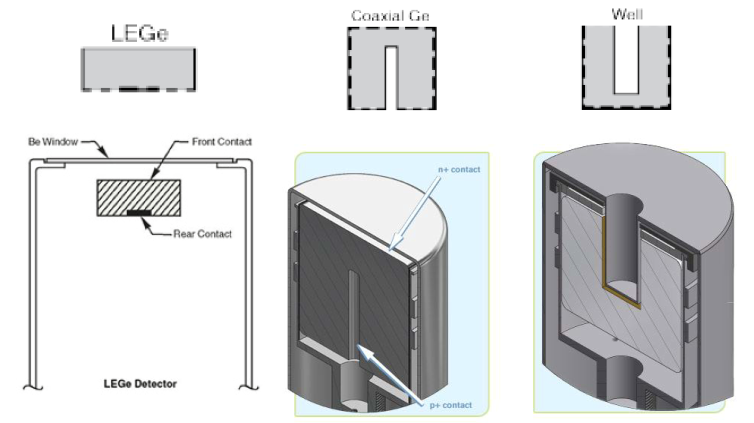
Germanium detectors have three types of shapes: planar, coaxial and well (Figure IX.6). Low-energy (LRGe) and broad energy-detectors (BEGe) are planar. The detector size in this construction mode is small and therefore these detectors are not able to efficiently detect high-energy gamma rays. In the coaxial mode the depletion layer is much thicker and therefore they are suitable in the detection of high-energy gamma rays. In the well-type the sample in placed inside the hole bored in the detector which considerably improves the counting efficiency.
Figure IX.6. Germanium detectors used for gamma spectrometry. Left: planar detector, middle: coaxial detector, right: well-type detector (http://www.canberra.com/products/detectors/germanium-detectors.asp).
The counting efficiency of the detectors is the fraction of gamma rays resulting in the formation of electric pulse of the total gamma ray number hitting the detector. The efficiency varies with detector type and the gamma ray energy as was shown in Figure IX.4. To compare efficiencies of various detectors this absolute efficiency is, however, not typically used but instead the efficiency is expressed in a relative manner by comparing the detector efficiency at 1332 keV photo-peak of 60Co to that of a Na(I) detector of size 3×3 inches at detector to source distance of 25 cm. This relative efficiency varies typically between 10% and 100%, the highest values obtained with larger detectors.
9.3. Energy calibration
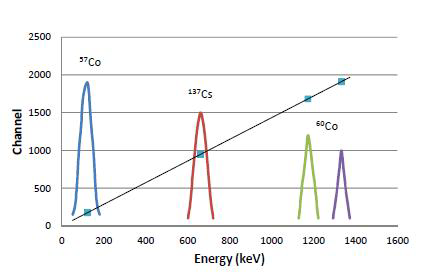
Multichannel analyzer sorts the pulses according to their heights, which are proportional to the energy of the gamma rays. To know what channel represents what energy the system needs to be calibrated. This is done by measuring standards of known energies depicted in Figure IX.7. In the figure the channel number are on the x-axis and the energies of the radionuclides on the y-axis. Here, three radionuclides with the following gamma energies are used:
| Nuclide | Energy |
| 57Co | 122 keV |
| 137Cs | 662 keV |
| 60Co | 1173 keV and 1332 keV |
By plotting a curve of the peak energy versus the channel where the mid point of the peak appears a calibration curve is obtained. This curve is linear since the initial pulses from detector, proportional to the energy of gamma rays, are amplified in a linear manner. For example, if the maximum of the 662 keV peak of 137Cs were in the channel 950, the maximum of the 122 keV peak of 57Co would be in the channel 175 (=950×122/662) and accordingly the 1173 keV and 1332 keV peaks of 60Co in channels 1683 (=950×1173/662) and 1911 (=950×1332/662). This linear calibration can now be used to identify unknown peaks in the spectrum. If, for example, a peak maximum of an unkonown sample was found in the channel 1198, one could see from the line that this channel corresponds to 835 keV energy. By examining spectrum library this energy could be shown to belong to 54Mn. Modern gamma spectrometers both store the calibration curve in their memory and also have a spectrum library and do the identification analysis automatically. They utilize not only gamma energies of each radionuclides but also relative intensities in case the nuclide has several gamma transitions.
One needs to bear in mind that the channels where each peaks go to depends on the settings of the amplifier: the higher the amplification the higher is the channel where peaks go. For example, when amplifier gain is doubled, the 662 keV peak of 137Cs would be found in the above mentioned case in which the channel is 1900 instead of 950.
Figure IX.7. Energy calibration in gamma spectrometry.
9.4. Efficiency calibration
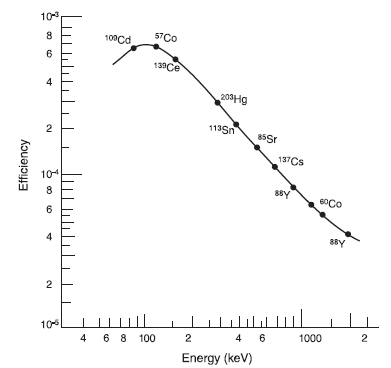
As is seen from Figure IX.4 the detector efficiency as a function of gamma ray energy is not constant but varies considerably. This must be taken into account by carrying out an efficiency calibration. The system is calibrated by measuring a mixture of radionuclides with a wide range of gamma photopeak energies. The activities of radionuclides should naturally be known and their values should be certified. Such radionuclide mixtures with certified activities are commercially available for efficiency calibration. At least seven radionuclides with varying energy should be used in the mixture. More radionuclides are needed for energy range below about 200 keV since the efficiency here varies in a more complex manner than at higher energies where an approximately linear relationship is obtained between energy and efficiency when presented in logarithmic scales. An example of composition of such standards with energy range from 60 keV to 1836 keV is presented in Table IX.I. This standard is meant for high accuracy calibration and consists of twelve nuclides. The standard is measured sufficiently long time to get at least 10000 counts to every photopeak and their net count rates are calculated by subtracting the background. Net count rates are then compared with activities to calculate the efficiencies and curve is fitted for the efficiencies as a function of gamma energy, i.e. efficiency calibration curve is plotted (Figure IX.8). This calibration curve can then be used to calculate the counting efficiency of photopeaks in actual sample measurements. Software of modern gamma spectrometers do this automatically based on the calibration curve stored in their memory.
Table IX.I. Composition of a standard for efficiency calibration of gamma spectrometer (NIST).
| Nuclide | Photopeak energy (keV) | Nuclide | Photopeak energy (keV) |
|---|---|---|---|
| 241Am | 59.5 | 85Sr | 514.0 |
| 109Cd | 88.0 | 137Cs | 661.7 |
| 57Co | 122.1 | 54Mn | 834.8 |
| 139Ce | 165.9 | 65Zn | 1115.5 |
| 203Hg | 279.2 | 60Co | 1173.2 and 1332.5 |
| 113Sn | 391.7 | 88Y | 1836.1 |
Figure IX.8. Efficiency calibration curve (http://www.canberra.com/literature /fundamental-principles/).
Efficiency calibrations are typically done in pure water solutions with the density of approximately 1 g/mL. Calibration curves are determined for all geometries used in actual sample measurements, i.e. for different sample vials, volumes and distances from the detector. When a liquid sample has an essentially different density than that of water, for example, in the case of solutions with high salt concentrations, self-absorption of gamma rays in the sample creates an additional challenge. This is more important with low energy gamma rays. For this kind of samples additional calibrations are needed to account for the density. Even more challenging is the calibration of solid samples due to the lack of proper solid standards with certified radionuclide activities. One can prepare own solid standards by mixing radionuclide standard solution with the solid matrix, sediment for example, and evaporating the solution. When using these kinds of in-house standards the composition of the actual samples should not essentially vary from that of the standard. Another way to do the efficiency calibration for solid samples is to use computational methods, for example by using Monte Carlo computer models. In these, the self-absorption is calculated by taking into account the density and elemental composition of the sample.
9.5. Interpretation of gamma spectra
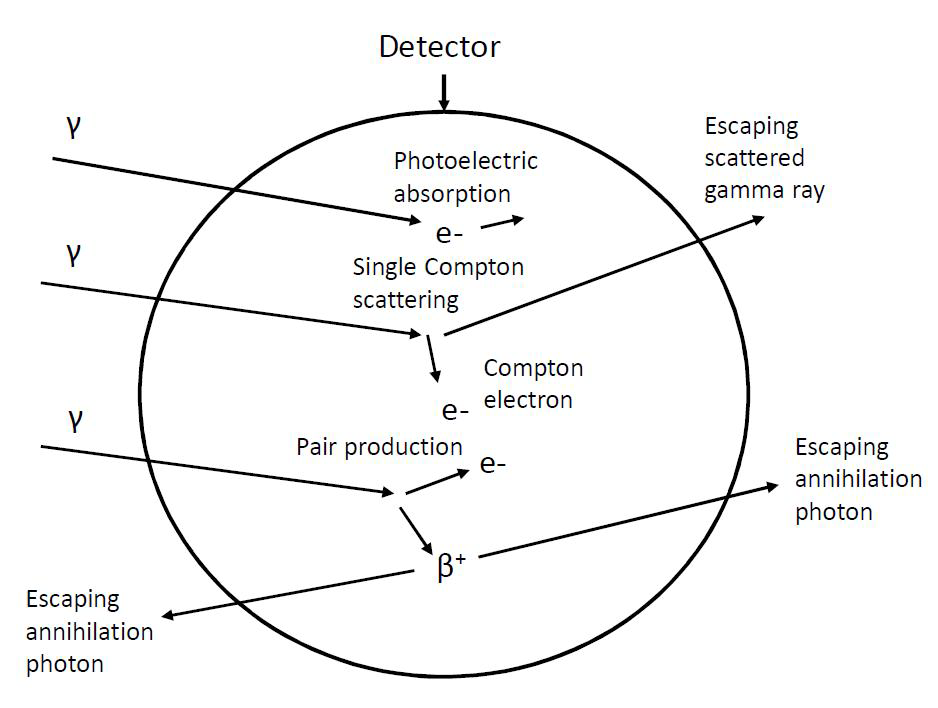
In the interpretation of gamma spectra all three major atomic scale interaction processes of gamma rays with detector material need to be taken into account. These are photoelectric effect, Compton effect and pair formation (Figure IX.9).
Figure IX.9. Photoelectric effect, Compton effect and pair formation in a gamma detector (circle) and escape of gamma rays from the detector.
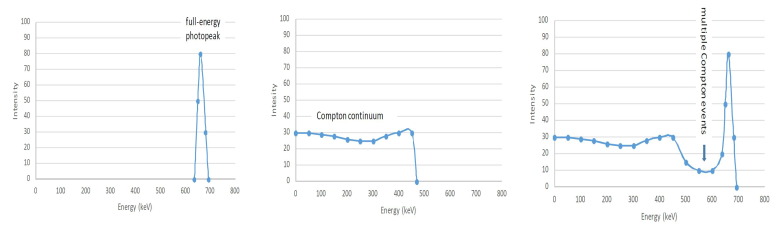
In the photoelectric effect a gamma ray loses its energy to a shell electron and these electrons create electric pulses of approximately same height. These can be seen as a peak in the gamma spectrum (IX.9, left side). Another area in the spectrum (IX.9, middle) is the Compton continuum, which is created when gamma ray loses only part of its energy to an electron and the scattered gamma ray escapes the detector. If Compton-scattered gamma ray will not escape the detector but loses its residual energy in a further photoelectron event the created total electric pulse will go to the photopeak area. Varying proportion of the gamma energy is lost to Compton electrons and therefore a continuum is seen. Compton electrons do not, however, have continuous energy between zero and the photopeak energy (Eγ) but their spectrum ends at about 200 keV less than the Eγ. This is due to fact that the maximum energy that the gamma ray can lose is when it is scattered to opposite direction to its initial path and the maximum energy of the scattered gamma ray in this case is about 200 keV less than its initial energy, more or less irrespective of the initial energy. Thus a valley is created between the Compton continuum and the photopeak. As seen from the right side of the Figure IX.10 there are, however, pulses in this valley. These are due to simultaneously occurring multiple Compton events and summation of the ensuing electric peaks. At part of such summation pulses go to the photopeak area and they need to be subtracted in the way later described.
Figure IX.10. Photopeak, Compton continuum and their combination in a gamma spectra.
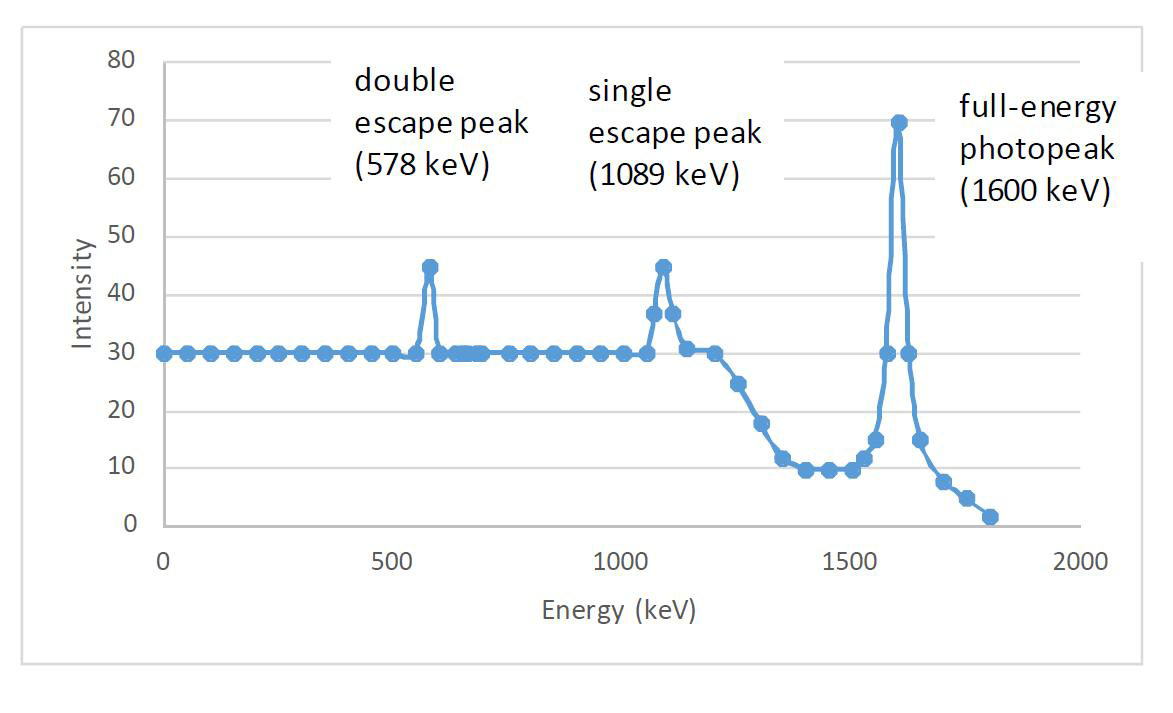
Gamma rays with energies higher than 1.022 MeV may undergo pair formation, i.e. turn into an electron and a positron. If they both lose their energy in the detector an electric pulse goes to the photopeak area. However, since the positron is not stable but annihilates after losing its kinetic energy with an electron to form two gamma rays of 0.511 MeV energy. In the case where one of these escapes the detector, a peak at Eγ - 0.511 MeV is created and correspondingly a peak at Eγ -1.022 MeV when both annihilation gamma rays escape (Figure IX.11).
Figure IX.11. Peaks appearing in a gamma spectrum due to pair formation and escape of annihilation gamma rays from the detector.
Still there may be additional peaks in gamma spectra. If two gamma rays simultaneously lose their energy in the detector a sum peak will be formed which is called coincidence summing. Furthermore, X-rays formed after electron capture, internal conversion and formation of Auger electrons may appear at the low energy region, but only when broad energy detector (BEGE) is used. In summary, gamma spectra are complicated, especially when several radionuclides are measured from same sample. Fortunately, there are computer programs, such as the SAMPO program, that take care of the peak analysis.
9.6. Subtraction of background
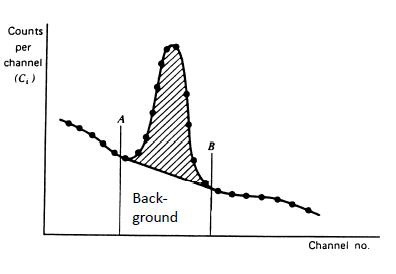
From gamma spectra radioactivities are determined from net peak areas of the photopeaks. In total peaks there are background counts created by external radiation, electric noise, Compton background of the radionuclides, if any, with higher photopeak energy and from multiple Compton events of the measured radionuclide. To get the net peak area the Compton background pulses are subtracted in the way presented in Figure IX.12. In addition, the pulses coming from external sources are subtracted from net peak area based on a separate background measurement but only if there is a peak, corresponding to the measured photopeak, in the background spectrum.
Figure IX.12. Subtraction of Compton background from gross photopeak area.
9.7. Sample preparation for gamma spectrometric measurement
Typically gamma spectrum is measured from samples without pretreatment by packing the sample into vial used in efficiency calibration. Also, the sample volume needs to correspond to a calibrated volume. Sometimes, however, pretreatment of samples is necessary. In cases where the activity concentration is so low that the activity of the target nuclide cannot be determined in a reasonable time, preconcentration is needed. For example, 137Cs concentration in natural waters is usually so low that even measuring one-liter samples does not allow its detection in a reasonable time. Thus 137Cs is preconcentrated by evaporation into a smaller volume or is chemically separated, for example, by precipitation with ammonium phosphomolybdate. The latter method also separates efficiently 137Cs from interfering radionuclides and thus gives a more accurate result.
textbook/nrctextbook/chapter9.1738336272.txt.gz · Last modified: 2025-01-31 16:11 by Merja Herzig
A-CINCH Consortium
email: mst@evalion.cz | tel: +420 224 358 331 | Copyright © 2021 A-CINCH
This project has received funding from the Euratom research and training programme 2019–2020 under grant agreement No. 945301.