User Tools
Sidebar
textbook:nrctextbook:chapter12
Table of Contents
12. Liquid scintillation counting
Chapter 12 from BASICS OF NUCLEAR PHYSICS AND OF RADIATION DETECTION AND MEASUREMENT - An open-access textbook for nuclear and radiochemistry students by Jukka Lehto
Liquid scintillation counting is primarily used to measure beta radiation (3H, 14C, 32P). It can, however, also be used for alpha radiation, low energy gamma- or X-rays, as well as measuring conversion- and auger-electron emitting samples. In addition, the liquid scintillation counters can be utilized in Cherenkov radiation measurement.
12.1. The principle of liquid scintillation counting
Liquid scintillation counting is based on the fact that the radioactive sample and a scintillator agent is dissolved into the same solvent. Three components thus comprise the measured sample: a radioactive sample, an organic solvent or solvent mixture, and one or more scintillation agents. The scintillation agent molecules, also called phosphors and fluors, entirely surround the decaying nuclide and thus avoids the harm of self-absorption and offers $4\pi$- counting geometry, in which all emitting particles or rays are detectable.
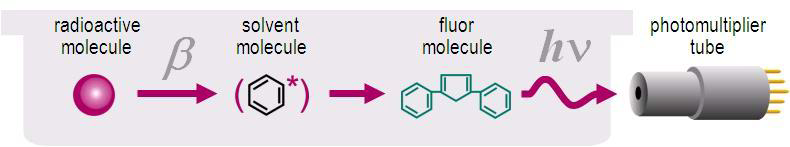
In the event of the decay of the nucleus the released beta particles collide with the solvent molecules, which are in the majority, and transfer their energy to them. These excited solvent molecules then release energy to other molecules. At some point, the energy is received by the scintillation molecules, which are able to release the excitation energy as light. Using a photomultiplier tube, these light pulses, lasting 3-5 ns, are changed into electrical pulses, their height is measured in an analyzer and registered to the different channels of the multichannel analyzer according to pulse height. The height of the pulse obtained by liquid scintillation counting is proportional to the original energy of the radiation and as the counters are equipped with a multichannel analyzer, they are suitable for energy spectrometry (Figures XII.1-4).
Figure XII.1. Functioning of the scintillation agent.
Several commercial scintillation liquid mixtures (scintillation cocktails) for liquid scintillation counting are available, containing both solvents and scintillation agents. Liquid scintillation measurements are generally done in either 20 ml or 6 ml plastic or glass vials, of which polyethylene bottles are the most common.
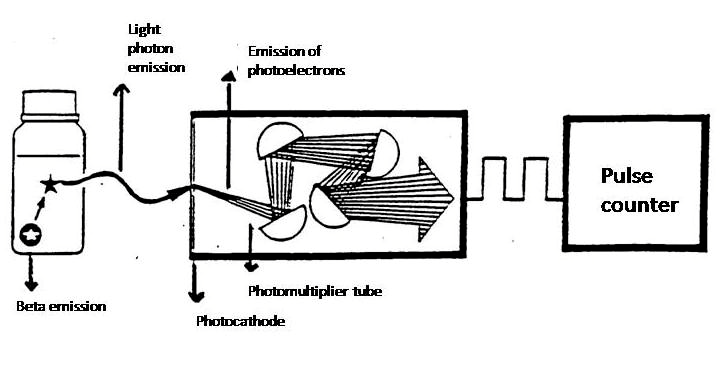
Figure XII.2. Principle of liquid scintillation counting (http://www.perkinelmer.com/ Resources/TechnicalResources).
Figure XII.3. The emergence of light pulses in liquid scintillation processes.
Figure XII.4. Light pulse detection.
12.2. Solvents
The solvent component of scintillation cocktail has two functions: it must be able to dissolve the sample and scintillation agent, as well as effectively transfer the energy from radioactive particle or ray to the scintillation agent. The best solvents are the aromatics such as xylene, toluene, benzene, and cumene. Aliphatic solvents, such as 1,4-dioxane and cyclohexane are also used. To improve the dissolution of the sample into the scintillation system, many secondary solvents are also used. The benefits of the newer liquid scintillation solvents, e.g. di-isopropylnaphthalene (DIN) and phenyl–o–xylylethane (PXE), are that they have a lower flammability, volatility and lack of odor, lower toxicity or irritancy, biodegradability, and a better solubility and counting efficiency.
12.3. Scintillation agents
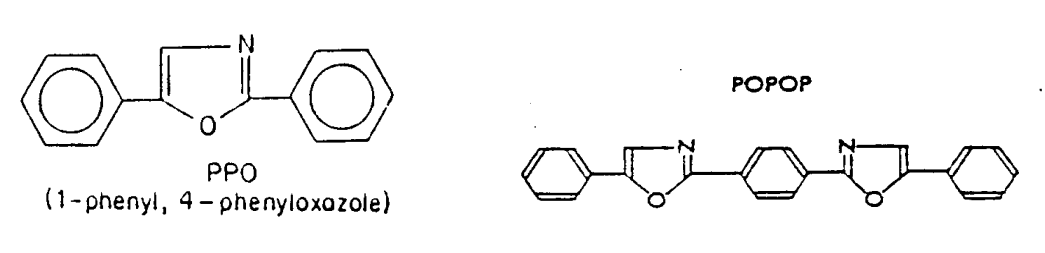
Tens of scintillators are recognized for use in liquid scintillation counting. Common ones are p-oligophenyls, or oxazole and oxadiazole compounds. The function of scintillators is to convert as much of the energy received via solvent molecules into light photons. The best scintillation materials have nearly 100% efficiency. Usually the scintillation material alone is not enough, because the sample may absorb light in the emitted wavelength range. In this case, a secondary scintillator is added to the liquid scintillation cocktail, i.e. spectrum transfer agents, which after excitation by the light of the primary scintillation emit longer wavelengths. Below are examples of primary scintillation material (PPO) and secondary scintillation material (POPOP).
Figure XII.5. Primary scintillation agent PPO (1-phenyl-4-phenyloxazole) and secondary scintillation agent POPOP.
12.4. Liquid scintillation counter
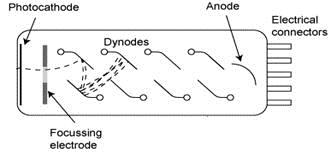
The basic element of the liquid scintillation counter is a photomultiplier tube (Figure XII.6), which transforms the light photons into electrons and amplifies them into measurable electrical pulses. At the front end of the photomultiplier tube that the photons hit is a photocathode typically made from Cs3Sb. When light photons hit the photocathode, it emits electrons. The electrons emitting in the photomultiplier tube are then amplified by dynodes, of which there are 10-14. Between successive dynodes is a voltage applied. The dynodes are also made of Cs3Sb and when the electrons hit them, the voltage causes the electrons to be amplified due to their growing kinetic energy. The voltage through the tube is 1000-2000V, which causes the electrons to be amplified by a factor of 106.
Figure XII.6. Photomultiplier tube (http://mxp.physics.umn.edu/s09/projects/S09_MuonEnergy/details_1.htm).
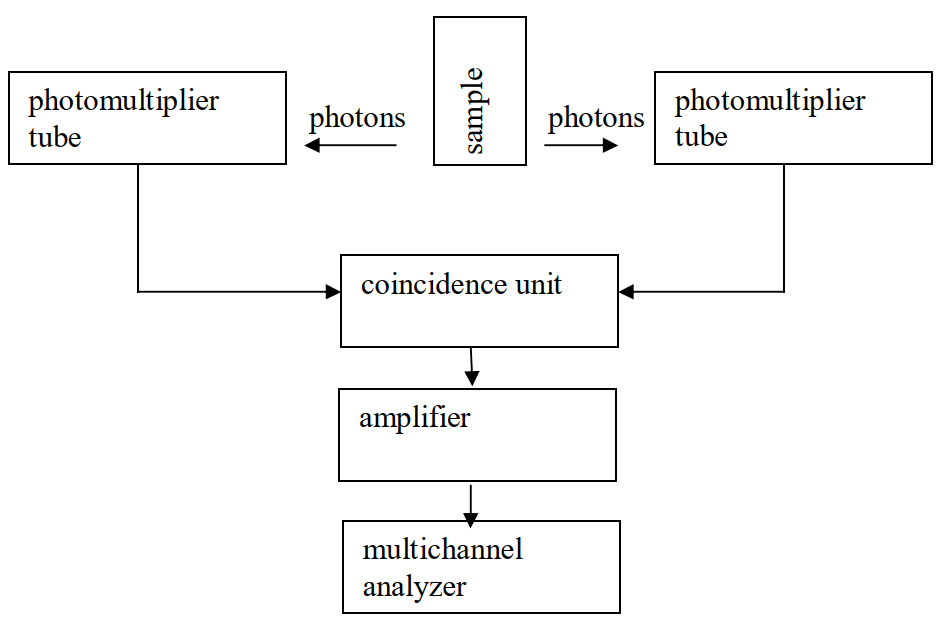
Liquid scintillation counting is used to detect light pulses with photomultiplier tubes using the coincidence technique (Figure XII.7). The sample is between two photomultiplier tubes, which are situated at an angle of 180º from each other. When the radionuclide decays in the scintillation cocktail, a large amount of light photons are simultaneously (in 10-9 s) generated and randomly emitted in every direction. The counter unit only registers pulses coming "simultaneously" (for up to 10-7 seconds) from the coincidence unit from both photomultiplier tubes and rejects single pulses coming from only one photomultiplier tube. The coincidence unit of the liquid scintillation counter is an electronic portal, which is open for 10-7 s at a time, in other words 100 times the duration of the pulse. With the aid of the coincidence technique the interfering effect of the single pulses is greatly reduced. In this way a lower background is achieved, when the electronic noise of the photomultiplier tubes, pulses from chemiluminescence and phosphorescence, as well as pulses from external radiation are nearly eliminated.
Figure XII.7. Liquid scintillation counter principle.
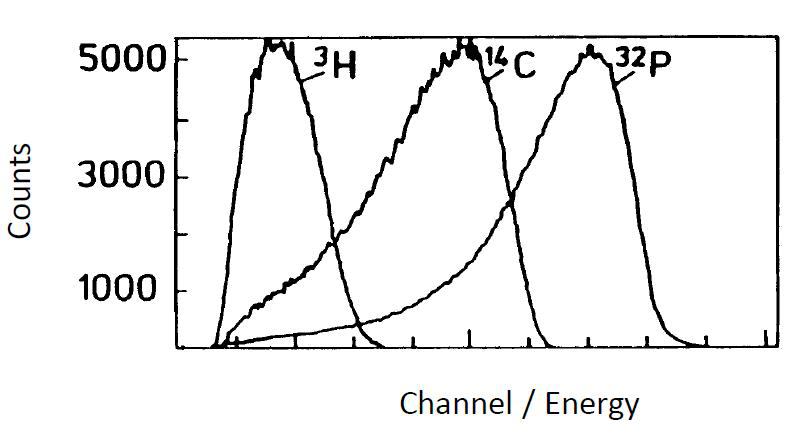
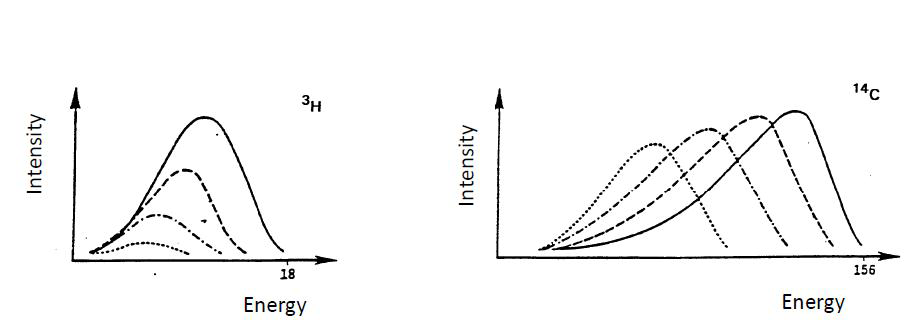
High count rates are able to be measured by a liquid scintillation counter, because the light pulse lasts only a very short time (10-9 s). If, for example, the sample activity is 106 Bq (which is so high that it is rarely measured), a decay occurs on an average every 10-6 second in a sample. This is 1000 times longer than the duration of a single light pulse and 10 times longer than a coincidence portal is open. Therefore, when measuring even such high activity each pulse can be detected individually without disturbance from the next pulse. From the coincidence unit the pulses go into a multichannel analyzer, which counts the pulses and differentiates them to different channels according to their height. The number of photons generated by liquid scintillation process is proportional to the initial energy of the beta particles. Tritium, for example, with a maximum energy of 18 keV, generates an average of 35 photons and 14C, with a maximum energy of 180 keV, an average 350 photons. As the photomultiplier tube amplifies pulses by a constant factor, the pulses coming into the analyzer are proportional to the energy of the beta particles. Since the energy distribution of particles generated in beta decay is continuous, a continuous spectrum, not a line spectrum, is obtained by the liquid scintillation counter. The measuring of alpha radiation, however, yields a line spectrum. The liquid scintillation counters show the spectrum of beta particle energies on a logarithmic energy scale, because their energies vary greatly. The figure below shows the individually determined liquid scintillation spectra of three nuclides (3H: Emax 18 keV, 14C: Emax 180 keV, 32P: Emax 1700 keV).
Figure XII.8. The individually determined liquid scintillation spectra of 3H (Emax 18 keV), 14C (Emax 180 keV) and 32P (Emax 1700 keV).
Since the spectra overlap, the simultaneous measuring of several beta emitters is difficult. The separation of two nuclide spectra is still reasonably simple, if their energies differ sufficiently; however, if a third nuclide is simultaneously being determined it becomes virtually impossible.
Liquid scintillation counters are equipped with an automatic sample changer. The samples are placed in either sample sites of a conveyer or of counter cartridges. One sample at a time is measured in a sealed lightproof counting chamber. The light emissions from the sample are gathered as efficiently as possible to the photocathodes of the photomultiplier tubes, which is why the counting chamber walls are aluminum mirrors or painted with titanium oxide.
12.5. Quenching
For determining the activity of radioactive samples, their count rates are often compared to those observed with a standard of known activity. Liquid scintillation counting also uses this approach. This approach, however, requires that both the unknown sample and the standard are measured entirely under the same conditions. In measuring beta radiation by liquid scintillation counting, the measurement conditions are rarely the same, because a varying amount of quenching occurs in the samples. Quenching means that either the beta particle energy is absorbed by the measurement sample (liquid scintillation cocktail) before it causes scintillation agent excitation and further light formation or the light emitted by the scintillation agent is absorbed in the sample, therefore being not registered as electrical pulses in a photomultiplier tube. The most difficult problem in liquid scintillation counting is resolving quenching and its impact. There are three types of quenching, all of which result in the detection of reduced count rates. In physical quenching the beta particle range does not extend to the scintillation agent, in chemical quenching the energy transmission efficiency from beta particle to the solvent and the scintillator is lowered, and in color quenching the photons are absorbed in the colored substances in the sample.
Quenching can be somewhat reduced by adding more scintillator, lowering the temperature, and using a scintillator with the shortest possible fluorescence time (the quenching agent does not have time to intercept the energy). Usually it must be accepted, however, that a sample has some quenching and its effect on the count rate found out by standardization. Some substances are particularly effective quenching agents even with a concentration of less than 1 ppm. The most common absorbing substance is oxygen, from air, dissolved in scintillation solutions. The strongest absorbing materials are, e.g. peroxides, acetone, pyridine, chloroform, carbon tetrachloride, methanol, ethanol, halogens, aldehydes, acids, bases, and heavy metals.
Figure XII.9 shows the effect of quenching on the observed beta spectra. All four samples on both sides have the same activity, but their quenching varies. The shifting of the spectrum to lower channels is due the reduction in intensity of single light pulses. The decrease in the height of the spectrum, in turn, is due to the growing portion of beta particles remaining completely unrecorded. Therefore, even if the activity of the samples is the same, the obtained count rate varies greatly depending on the quenching. Thus, the observed count rates cannot be directly compared to those of the standards to allow direct calculation of the unknown sample activity until quenching is accounted for. This is accomplished by determining counting efficiency individually for each sample, which is the ratio of the observed count rate of the sample to the activity of the sample. Thus when the count rate ($\text{R}$) and the counting efficiency ($\text{E}$) are measured the activity ($\text{A}$) of the sample can be calculated by:
$$A = \frac{R}{E}$$
[XII.I]
Figure XII.9. The effect of quenching on the beta spectrum in liquid scintillation counting.
12.6. Methods for determining counting efficiency
Since quenching varies from one sample to another, the counting efficiency ($\text{E}$) of each sample must be determined, in order to calculate the activity ($\text{A}$, dpm) from the observed the count rate ($\text{R}$, cpm):
$$E(\%) = \frac{R(\text{cpm})}{A(\text{dpm})} \times 100\%$$
[XII.II]
When the count rates are corrected by the counting efficiency to get activity, they may then be compared with each other. The counting efficiency can be determined by many methods, three of which are described here: the use of an internal standard, the external standard channel ratio method, and the external standard end point method.
12.6.1.The use of an internal standard
Using the internal standard is the most accurate, but tedious. The sample is measured twice: first as it is and then by adding a known amount of the same nuclide as was in the sample and measuring again. Count rate growth is measured, and by comparing it to the amount of added activity the counting efficiency can be obtained as follows:
$$E(\%) = \left( \frac{\text{cpm}_2 - \text{cpm}_1}{\text{dpm}} \right) \times 100\%$$
[XII.III]
where $\text{cpm}_1$ = count rate of the sample $\text{cpm}_2$ = sum count rate of the sample and the added activity $\text{dpm}$ = amount of added activity $\text{E}$ = counting efficiency
The count rate of the unknown sample is then divided by the counting efficiency to get its activity, $A = \frac{{cpm_1 \times 100}}{{E(\%)}}$.
12.6.2. The external standard ratio method
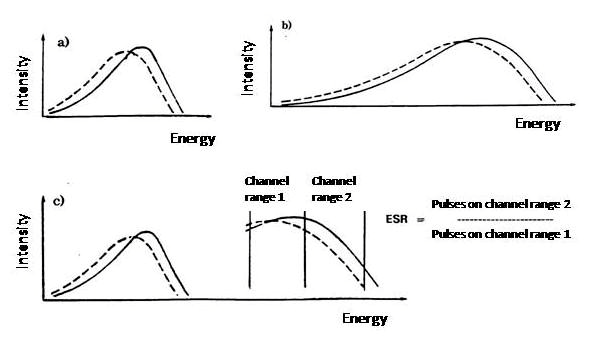
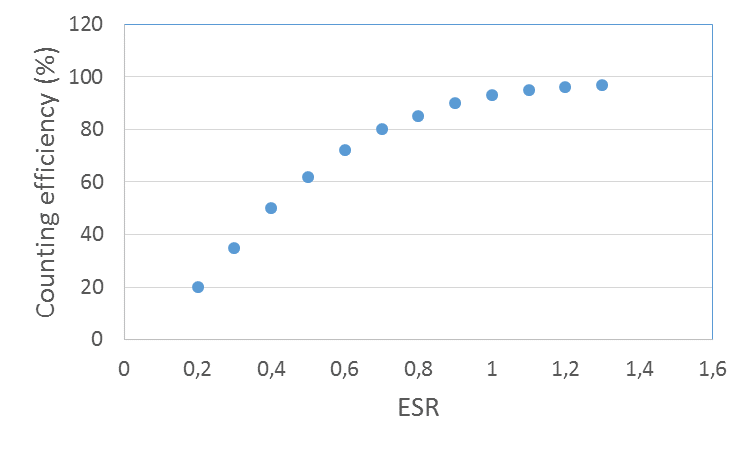
In the external standard ratio standardization method the device uses an external 226Ra source, which has an activity of about 400 kBq (10 μCi). In standardization step the Ra-source automatically rises next to the sample bottle in the measuring chamber, so that the gamma rays emitting from it also hit the scintillation cocktail. Compton electrons are generated when the gamma rays are absorbed into scintillation cocktail causing a spectrum similar to that of a beta particle emitting sample, only at a higher channel range (Figure XII.10). The pulses move towards lower channels as the quenching increases. The samples are measured twice: when measuring the actual sample the 226Ra source is not in the measuring chamber but is protected, while in standardization it is brought next to the sample bottle. The pulses caused by the radium standard are divided into two channel ranges and the pulse number ratio of these channel ranges is calculated. This external standard ratio (ESR) is proportional to quenching: the more quenched the sample, the more pulses move to the lower channels, in other words, the lower is the external standard ratio. Accordingly, as the sample is quenched the counting efficiency is also reduced.
For the standard curve, which is called the quenching curve, a series of samples are measured (quenching series), all of which have the same activity for a particular nuclide, but the quenching is varied, for example, by adding an increasing amount of CHCl3. The quenching increases with this addition, while the counting efficiency, as well as the ESR, decreases. The counting efficiency ($\text{E}$ (%)), i.e. the count rate in the channel range covering pulses of an unquenched sample divided by the sample activity is then plotted on the curve as a function of the external standard ratio (ESR) (Fig. XII.11). When an unknown sample is then measured, first the external standard ratio is determined and by using this value the counting efficiency, e.g. 70%, is read from standard curve. The sample activity is then calculated by dividing the determined count rate by the counting efficiency (e.g. 0.70). The standard curve is in practice stored in the memory of a liquid cintillation counter and device does the calculation automatically.
Figure XII.10. a) unquenched sample (—–) and quenched (- - -) spectrum; b) spectra caused by external standard: unquenched (—–) and quenched (- - -) spectrum; c) external standard ratio (ESR) calculation principle.
12.6.3. External standard spectrum endpoint method
The measure of quenching in the external standard spectrum endpoint method, is as the name implies, the endpoint of spectrum caused by the external standard: the greater the quenching, the lower the channel on which the spectrum ends. Since it is difficult to exactly define the endpoint of the spectrum, the endpoint is determined by the channel under which 99.5% of all of the pulses occur. Just as in the sample and external standard channel ratio methods, the external standard endpoint, SQP-value, is determined for the quenching series as a function of counting efficiency and the obtained quenching curve is used to calculate the activity of unknown samples.
12.7. Cherenkov counting with a liquid scintillation counter
When a charged particle passes through the medium at a speed faster than light, it polarizes the medium molecules. When this polarization is released, the medium molecules emit photon radiation of ultraviolet and visible light spectrum range. This phenomenon, which is called Cherenkov radiation, can be used for beta radiation measurement because it is also identifiable by the photomultiplier tube of a liquid scintillation counter. Beta particle energy must be at least 263 keV for Cherenkov radiation to occur in water. In practice, Cherenkov radiation is only useful for beta radiation measurement when the beta radiation energy is at least 800 keV. For Example, only 2% of counting effectiveness is achieved in Cherenkov counting with 137Cs (average beta energy of 427 keV), while 25% counting effectiveness is achieved with 32P (average beta energy of 695 keV).
Cherenkov radiation measurement has some important advantages compared to liquid scintillation counting. First, larger amounts of the solution can be measured since no liquid scintillation solution needs to be added to the counting vial. Second, no costly liquid scintillation waste is generated in Cherenkov counting.
12.8. Alpha measurement with a liquid scintillation counter
The determination of alpha_emitters by liquid scintillation counter is a very convenient method. The sample preparation is considerably simpler than when measuring with semiconductor detectors. For measurement with semiconductor detectors the sample must be very thin, i.e. "massless", so that the alpha radiation is not absorbed into the sample. In liquid scintillation counting, this is not generally a problem, because alpha-emitting radionuclides mixed with liquid scintillation cocktail are in immediate contact with the scintillator. Since the energies of alpha particles are high, generally 4-6 MeV, in practice their detection efficiency is nearly 100% and quenching is usually not a problem. In addition, because the liquid scintillation counters have sample changer, its measurement capacity is superior to that of the semiconductor. The disadvantage that liquid scintillation counting has compared to the semiconductor detectors is its significantly worse energy resolution. The best semiconductor detectors will yield a peak width values at half maximum of 10-20 keV, while liquid scintillation counters get, at best, only 200 keV. Therefore, alpha energies that are close to each other are not able to be measured separately with a liquid scintillation counter. Another problem in measuring alpha radiation with a liquid scintillation counter h when measuring environmental samples is the fact that the beta radiation forms a high background that interferes with the measurement. Today, however, there are liquid scintillation counters capable of differentiating between the pulses caused by alpha particles from those caused by beta particles. The electric pulse induced by beta particles is considerably shorter, around a few nanoseconds, than the alpha particle induced pulse that lasts several tens of nanoseconds. Below is a spectrum, in which there are alpha peaks of 226Ra and its daughters, and the beta spectrum of 226Ra daughter nuclides separated by the pulse shape analysis.
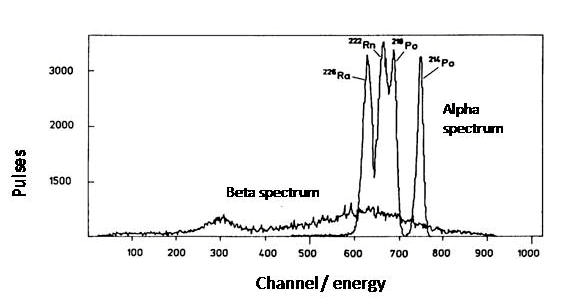 Figure XII.12. The alpha- and beta spectra of 226Ra and its daughters obtained by pulse shape analysis.
Figure XII.12. The alpha- and beta spectra of 226Ra and its daughters obtained by pulse shape analysis.
12.9. Sample preparation for liquid scintillation counting
In addition to the determination of the counting efficiency there is a second critical task in the liquid scintillation counting: preparation of samples. Whenever possible, a homogeneous measurement sample should be obtained in which the radionuclide is evenly dissolved in the liquid scintillation cocktail. If the sample is an organic solvent, it is usually directly soluble in the liquid scintillation cocktail. This, however, is rarely the case. Usually the samples for measurement are aqueous samples. Water is only partially soluble in organic liquid scintillation solvents, but even the best cocktails can reach as high as 50% water concentration. Water samples can also be measured as gels, in which case the water is evenly distributed in the liquid scintillation cocktail. Many insoluble organic substances must be decomposed before measurement. Dissolution can be done with e.g. perchloric-hydrogen peroxide oxidation or burning the sample and collecting the CO2 for measuring if 14C is to be measured. If 3H is to be measured, then H2O is collected. PerkinElmer offers an automatic system, Sample Oxidizer, where the organic sample is decomposed with a flame and tritium is collected as water into another scintillation vial and radiocarbon to another vial after conversion into a carbamate in Carbosorb column. Liquid scintillation cocktail is then added to the both vials - automatically too (Figure XII.13). Insoluble samples, e.g. fine solids and chromatography masses can be measured as heterogenetic samples by adding them and a liquid scintillation cocktail to a gelling substance, like aluminum stearate, forming a gel in which the precipitate is evenly distributed. Radioactive chromatography or electrophoresis strips can be measured directly by immersing them in a liquid scintillation cocktail containing flask. The sample preparation methods are summarized in the Figure XII.14.
 Figure XII.13. Perkin Elmer Sample Oxidizer to prepare 3H and 14C samples for liquid scintillation counting after decomposition of organic samples (http://shop.perkinelmer.com/Content/applicationnotes/app_oxidizercomparisonsampleoxidation.pdf).
Figure XII.13. Perkin Elmer Sample Oxidizer to prepare 3H and 14C samples for liquid scintillation counting after decomposition of organic samples (http://shop.perkinelmer.com/Content/applicationnotes/app_oxidizercomparisonsampleoxidation.pdf).
Figure XII.14. Sample preparation methods for liquid scintillation counting.
textbook/nrctextbook/chapter12.txt · Last modified: 2025-04-28 11:17 by Merja Herzig
A-CINCH Consortium
email: mst@evalion.cz | tel: +420 224 358 331 | Copyright © 2021 A-CINCH
This project has received funding from the Euratom research and training programme 2019–2020 under grant agreement No. 945301.