User Tools
Sidebar
textbook:nrctextbook:chapter15
Table of Contents
15. Nuclear reactions
Chapter 15 from BASICS OF NUCLEAR PHYSICS AND OF RADIATION DETECTION AND MEASUREMENT - An open-access textbook for nuclear and radiochemistry students by Jukka Lehto
A nuclear reaction is an event in which a nucleus is targeted by a projectile (proton, deuteron, alpha particle, etc.) or gamma ray causing one of the following interactions with the nucleus:
- Transmutation or the formation of a new nucleus
Since the specific aim of this communication is production of new nuclei, primarily radionuclides, in nuclear reactions, transmutation lead reactions will be the focus. Scattering is mentioned only briefly. Projectiles can be very heavy, even uranium nuclei, but this communication will concentrate on lighter ones (p, d, α, n).
In nature, nuclear reactions occur in the following sources:
- The cosmic protons and alpha particles of the upper parts of the atmosphere (as well as the neutrons generated by these nuclear reactions) cause nuclear reactions upon hitting with the gas molecules of the atmosphere. The most important of these is the emergence of the radiocarbon,14C, from atmospheric nitrogen.
- Fusion reactions occuring in stars.
- A very small number of neutron activation reactions caused by neutrons generated in spontaneous fission of uranium in bedrock and overburden.
The first artificial nuclear reaction was achieved by Ernst Rutherford in 1919, when he targeted nitrogen gas with alpha radiation generated in 214Po decay, which resulted in the following reaction:
$${}^{14}\mathrm{N} + {}^{4}\mathrm{He} \rightarrow {}^{17}\mathrm{O} + {}^{1}\mathrm{H}$$
[XV.I]
The products of the reaction were oxygen, its 17O isotope and protons. The above reaction equation can also be presented in a shorter form:
$${}^{14}\text{N} ({}^{4}\text{He}, p) {}^{17}\text{O}$$
or
$${}^{14}\text{N} (\alpha, p) {}^{17}\text{O}$$
[XV.II]
where the left side of the parentheses is the target nucleus and on the right is the result nucleus. Inside the parentheses on the left is a projectile particle/ray and on the right an emitting particle/ray. The bombarded substance is called the target. The neutrons needed for nuclear reactions are primarily obtained by research reactors, but also in neutron generators. The positive projectile particles are produced by particle accelerators that are e.g. the van de Graaf accelerator, linear accelerator, cyclotron, and synchrotron.
The first particle accelerator, which could achieve nuclear reactions, was developed in the early 1930s. The first nuclear reaction that created an artificial radionuclide was accomplished in 1934, when Frederick Joliot and Irene Curie used a particle accelerator to bombard aluminum with alpha particles and produced 30P, which is a positron emitter. The reaction was, therefore, ${}^{27}\text{Al} (\alpha, p) {}^{30}\text{P}$.
15.1. Nuclear reaction types and models
In most nuclear reactions the target nucleus absorbs the projectile particles (p, d, α, n) and emits other particles in a fairly short period of time (10-14-10-18 s). A liquid drop model was developed to describe these types of reactions. The kinetic energy of the projectile particle and its merging with the nucleus generates bonding energy that spreads evenly as the nuclear excitation energy. Much like a liquid droplet, the particles at some point in the nucleus have such an ample energy that it "evaporates" from the nucleus. The liquid drop model is supported, for example, by the fact that the reactions
$${}^{12}_{\phantom{1}6}\text{C} + {}^{144}_{\phantom{1}60}\text{Nd} \rightarrow {}^{150}_{\phantom{1}66}\text{Dy} + 6n$$
and
$${}^{20}_{10}\text{Ne} + {}^{136}_{\phantom{1}56}\text{Ba} \rightarrow {}^{150}_{\phantom{1}66}\text{Dy} + 6n$$
[XV.III]
have roughly equal probability as a function of the projectile energy (i.e. excitation function, which is explained later). In this case, the explanation is that an excited intermediate nucleus or compound nucleus ${}^{156}_{\phantom{1}66}\text{Dy}$ is generated in both cases, which breaks down in the same way, regardless of its origin.
The liquid drop model is not able to explain all of the observed reactions, in particular those with high projectile energy. These reactions are, for example, spallation reactions, in which a large number, even many tens of nucleons, are emitted from the nucleus and the resulting nucleus is of a clearly lighter element. Such reactions are also the fragmentation reactions, in which instead of one several lighter nuclei develop. These reactions are depicted by the direct interaction model, according to which the intermediate nucleus does not have time to form, in other words the excitation energy has no time to spread throughout the nucleus and instead breaks down immediately as a direct effect of the projectile particles.
Fission is a reaction in which a heavy nucleus, e.g. 235U, decays into two lighter elements. It is also explainable by the liquid drop model. Fusion is the opposite reaction to fission: lighter elements join together to form heavier nuclei.
Scattering is characteristic of neutron interactions with target nuclei. In scattering the projectile particle and emitting particle are identical. If there is no change in the energy of the nucleus during scattering, it is called elastic. In inelastic scattering the nucleus becomes excited and the kinetic energy of the emitting particle reduces.
15.2. The Coulomb barrier
When a positively charged particle (proton, deuteron, alpha particle etc.) collides with a nucleus, it is subjected to the positively charged protons of the target nucleus causing a repellent force. In order to penetrate into the nucleus the particle must have enough kinetic energy to overcome this repulsion, in other words cross the Coulomb barrier. The Coulomb barrier is higher, the greater the target substance atomic number is. The Coulomb repulsion in also dependent on the bombarding particle charge: it is larger for alpha particles that have a charge of +2 than protons, with a charge of +1. According to the Coulomb’s law the repulsion force is
$$F_{\text{coul}} = k \cdot e \cdot Z_1 \cdot e \cdot Z_2 / x^2 = k \cdot e^2 \cdot Z_1 \cdot Z_2 / x^2$$
[XV.IV]
where $\text{k}$ is Coulomb’s constant ($8.99 \times 10^9 \, \text{N} \, \text{m}^2 \, \text{C}^{-2}$), $\text{e}$ is the electron charge, $\text{Z}_1$ is the elemental number of the projectile particle, $\text{Z}_2$ is the corresponding value for the target nucleus, and $\text{x}$ is their distance. i.e. distance of their center points. So, this is the energy of projectile needed to cross the Coulomb barrier. Figure XV.1 shows the dependence of the Coulomb barrier on the nuclear charge of both the projectile particle and the target nucleus.
 Figure XV.1. The height of the Coulomb barrier for three projectile particles as a function of the target nucleus atomic number.
Figure XV.1. The height of the Coulomb barrier for three projectile particles as a function of the target nucleus atomic number.
15.3. Energetics of nuclear reactions
As in radioactive decay, the change in energy occurring in nuclear reactions can be calculated from the masses of the initial and resulting nuclei. The change in mass in a nuclear reaction $A(x,y)B$ is thus:
$$\Delta m = m_B + m_y - m_A - m_x$$
[XV.V]
The reaction energy $Q \, (\text{MeV}) \, \text{is} \, -\Delta m \, (\text{amu}) \times 931.5 \, \text{MeV/amu}$. If the mass in the reaction decreases, it means that the energy is released, i.e., it is an exoergic reaction. On the other hand, if the mass increases, it is an endoergic reaction. For example, in the equation [XV.II] for the reaction ${}^{14}\text{N} (\alpha, p) {}^{17}\text{O}$, the reaction $\text{Q}$ is
$$Q = 931.5 \, \text{MeV/amu} \left( 16.999131 + 1.007825 - 14.003074 - 4.002603 \right) \, \text{amu}$$ $$= 931.5 \, \text{MeV/amu} \times -0.001279 \, \text{amu} = -1.19 \, \text{MeV}$$
[XV.VI]
The total mass increases in this reaction by 0.001279 amu and thus this is an endoergic reaction, i.e. the kinetic energy of the projectile particle must import the required energy (1.19 MeV) to the target nucleus. In addition to this energy, the projectile particle must have enough energy to also encompass the kinetic energy of the emitting particle and the recoil energy of the resulting nucleus. The smallest possible projectile particle energy able to cause an endoergic reaction is called the threshold energy ($\text{E}_{th}$) of the reaction. From the energy and momentum conservation laws one can derive threshold energy value by:
$$E_{\text{th}} = \frac{m_x + m_A}{m_A} \times (-Q)$$
[XV.VII]
The threshold energy of the reaction ${}^{14}\text{N} (\alpha, p) {}^{17}\text{O}$ is thus $-(-1.19 \, \text{MeV}) \left( \frac{4+14}{14} \right) = 1.53 \, \text{MeV}$, being 0.34 MeV larger than the reaction energy.
To achieve an exoergic reaction, the projectile particle must have enough energy to cross the Coulomb barrier. In exoergic reactions the kinetic energy of the emitting particle is, however, not the same as the reaction energy since also in this case the resulting nucleus gets part of the released energy as recoil energy.
In addition to particle emission the nucleus also often emits gamma rays. For example, in the reaction
$${}^{184}_{\phantom{1}74}\text{W} + \alpha \rightarrow {}^{186}_{\phantom{1}76}\text{Os} + 2n$$
[XV.VIII]
the intermediate nucleus ${}^{188}_{\phantom{1}76}\text{Os}$ is generated, with an excitation energy of 20 MeV. The release of each neutron reduces the binding energy by 6 MeV and their kinetic energy of 3 MeV, i.e. a total of 18 MeV of excitation energy is removed with their emission. The remaining 2 MeV is not enough to overcome the binding energy of a third neutron, but this portion departs the nucleus as gamma radiation.
15.4. Cross sections
The cross section describes of the probability of a nuclear reaction occurrence. In other words, it tells us how large a fraction of bombarding particles brings about a nuclear reaction. The cross section is derived in this section.
Let’s expose a target with $\text{N}$ number of nuclei per unit volume ($m^3$) and $\text{dx}$ in thickness ($m$) with a coherent particle flux. When the target is so thin that the particle flux density $\phi_o$ (particles/m2×s1) does not essentially change, the decrease in particle flux density equals with the number of collisions leading to nuclear reactions in a unit of time per unit area ($m^2$):
$$- d\phi = \phi_0 \times N \times \sigma \times dx$$
[XV.IX]
where $\sigma$ is the probability of events or the cross section. When the target is so thick ($x$) that the particle flux decreases significantly, its value can be calculated with the equation XV.X, which is obtained by integrating the equation XV.IX. with respect to thickness.
$$\phi = \phi_0 \times e^{-\sigma \times N \times x}$$
[XV.X]
The unit of the cross section derived from the equation XV.VII is unit area. Because this has a very small value, barn (b = 10-28 m2, approximately a unit cross section of a nucleus) is used instead m2. If we neglect Coulombic interactions and nuclear forces the cross section should approximately be comparable with the size of a nucleus, which in fact applies to many neutron-induced reactions, i.e. cross sections are close to 1 barn. However, due to action of the repulsive and attractive forces from nuclear and Coulombic interactions the cross sections vary several orders of magnitude, both above and below 1 barn.
The decrease in particle flux in the target does not yet explicitly describe the number of a specific nuclear reaction. Several nuclear reactions can occur simultaneously in the target, for example a reaction can lead to the emission of one neutron and a reaction with emission of two neutrons simultaneously. When all simultaneous reactions are considered, the cross section is called the total cross section while when individual reactions are considered separately it is called a partial cross section in which case the total cross section is the sum of all the partial cross sections of simultaneous nuclear reactions.
15.5. Nuclear reaction kinetics
When nucleus B is produced by irradiating a nucleus A in a nuclear reaction, the equation for the growth rate of the resulting nucleus is:
$$\frac{dN_B}{dt} = \sigma \times \phi \times N_A$$
[XV.XI]
where $\text{N}_A$ and $\text{N}_B$ are the numbers of target and product atoms, respectively. If the product nuclide B is radioactive, it decays at the same time by a factor $-\lambda N_B$, where $\lambda$ is the decay constant of nuclide B. In this case, the total rate of growth for the product nucleus is
$$\frac{dN_B}{dt} = \sigma \times \phi \times N_A - \lambda N_B$$
[XV.XII]
When the irradiation time is t, the number of product nuclei is calculated at the end of irradiation by the formula XV.XIII, which is obtained by integrating the formula XV.XII in the time interval $0 - t$ assuming that at the start of irradiation $N_B = 0$.
$$N_B = \frac{\sigma \times \phi \times N_A}{\lambda} \times (1 - e^{-\lambda t})$$
[XV.XIII]
When producing radionuclides, the activity of the nuclide is of more interest than the number of nuclei. Since $A = \lambda \times N_B$, we can replace $N_B$ with $\frac{A}{\lambda}$ in the formula XV.XIII to get:
$$A_B = \sigma \times \phi \times N_A \left(1 - e^{-\lambda t}\right)$$
[XV.XIV]
The mass m is used instead of the target nuclei number $N_A$ and the half-life t½ is used instead of the decay constant $\lambda$, in which case the formula becomes
$$A_B = \frac{m \times I \times 6.023 \times 10^{23} \times \sigma \times \phi}{M} \times \left(1 - e^{-\ln 2 \times \frac{t}{t_{1/2}}}\right)$$
[XV.XV]
where $m$ is the mass of the target element, $I$ the target nuclide’s isotopic abundance of the element, 6.023·1023 is the Avogadro´s number, and $M$ is the molar mass of the element.
Figure XV.2. shows the relative amount of nuclide produced in the target as a function of irradiation time. Time here is the irradiation time divided by the nuclide’s half-life, i.e. it is the number of half-lives. As seen, 50% of the maximum obtainable activity (saturation activity) is produced during one half-life, 75% during two half-lives, and about 99% during ten.
 Figure XV.2. The relative amount of a radionuclide in the target as a function of irradiation time up to ten half-lives of the product nuclide and the decay of the product nuclide after irradiation.
Figure XV.2. The relative amount of a radionuclide in the target as a function of irradiation time up to ten half-lives of the product nuclide and the decay of the product nuclide after irradiation.
Finally, accounting for the continuing decay of the radionuclide after irradiation, the activity of the produced radionuclide can be calculated at the time point t* after the end of the irradiation by the formula
$$A_B = \frac{m \times I \times 6.023 \times 10^{23} \times \sigma \times \phi}{M} \times \left(1 - 2^{-\frac{t}{t_{1/2}}}\right) \times 2^{-\frac{t*}{t_{1/2}}}$$
[XV.XVI]
15.6. Excitation function
The probability of nuclear reactions is also dependent on the projectile particle energy. Presenting the cross sections of all individual reactions as a function of the projectile energy is called an excitation function. The Figure XV.3 demonstrates the excitation function of 54Fe irradiation with alpha particles. As seen, the predominant reactions at low projectile energy lead to emission of a single particle ($n$ and $p$). As the projectile energy increases also two particle emissions occur and at even higher energies also three particle emissions. The reactions in the figure are:
- $^{54}\text{Fe}(\alpha,p)^{57}\text{Co}$
- $^{54}\text{Fe}(\alpha,n)^{57}\text{Ni}$
- $^{54}\text{Fe}(\alpha,pn)^{56}\text{Co}$
- $^{54}\text{Fe}(\alpha,2n)^{56}\text{Ni}$
- $^{54}\text{Fe}(\alpha,2pn)^{55}\text{Fe}$
- $^{54}\text{Fe}(\alpha,p2n)^{55}\text{Co}$
Figure XV.3. The excitation function in irradiation of 54Fe with alpha particles (data from Houck and Miller, Physical Review 123(1961)231.
15.7. Photonuclear reactions
gamma radiation can cause nuclear reactions resulting in particle emissions, e.g. ($\gamma, n$) and ($\gamma, p$). These type of reactions are called photonuclear reaction. These reactions require a certain threshold energy from gamma rays to overcome the binding energies of protons and neutrons. The threshold energies of protons are higher than those of neutrons, because their removal from the nucleus also requires energy to cross the Coulomb barrier. Threshold energies are usually at least 5 MeV, so most of the gamma rays generated in radioactive decay do not cause nuclear reactions. Some nuclei, such as 2H, 9Be, and 13C, have lower threshold energies, however, and for example the 9Be($\gamma, n$)8Be reaction can already occur at gamma energies of 1.67 MeV.
15.8. Neutron induced nuclear reactions
Since neutrons have no charge they are not affected by the repulsion caused by the positively charged nuclei. Therefore, neutron induced reactions do not have a threshold energy. On the contrary, reactions are achievable even with very little energy. In fact, the reaction probability (cross section) for neutrons with energy less than 1 MeV is higher the lower the energy: the cross section is inversely proportional to the kinetic energy of the neutron (Figure XV.4.).
Figure XV.4. The excitation function of nuclear reactions induced by neutrons (neutron capture) in a 113Cd target http://thorea.wikia.com/wiki/Thermal,_Epithermal_and_Fast_Neutron_Spectra).
For slowest neutrons, called thermal neutrons having energies of 0.005-0.1 eV, the neutron capture cross sections are as high as 105 b. The ability of slow neutrons to cause nuclear reactions is due to their high wavelength of about 0.1 nm, while the wavelengths of faster neutrons (<0.1 MeV) is a thousand times smaller. Therefore, the probability of slower neutrons hitting the nuclei is greater.
Slow neutrons often cause a capture reaction, that is, the neutrons are absorbed into the nucleus and gamma rays are emitted. These gamma rays get kinetic energy from the binding energy of absorbed neutrons. Typically, the energies of the gamma rays are high, between 5-8 MeV.
As shown in Figure XV.4 the excitation function of neutrons at intermediate energies has many peaks. These are resonances, in which the excitation energy produced by the projectile particle nuclei are equivalent to the excitation levels of the nucleus. When the projectile energy value rises above 1 MeV the resonance states overlap and the excitation function levels.
Neutrons with a higher projectile energy can also produce reactions leading to alpha and proton emissions. Since their departure from the nucleus requires crossing the Coulomb barrier, these reactions always have a threshold energy.
Neutrons also cause fission reactions, which will be discussed in the next section.
15.9. Induced fission
Chapter V dealt with spontaneous fission and found that the reason for it is that a nucleus is too heavy and that it occurs only in the heaviest nuclei. In many ways, induced nuclear fission is similar to spontaneous fission. In both cases the nucleus disintegrates into two lighter nuclei, but not spontaneously in case of induced fission but through the excitation energy of external particles, typically neutrons, for example
$${}^{235}\mathrm{U} + \mathrm{n} \rightarrow ({}^{236}\mathrm{U})^* \rightarrow {}^{143}\mathrm{Ba} + {}^{89}\mathrm{Kr} + 3{}^{1}\mathrm{n} + 200\mathrm{MeV}$$
[XV.XVI]
where $(^{236}\text{U})^*$ is the excited intermediate uranium nucleus generated by neutron absorption, which quickly breaks down.
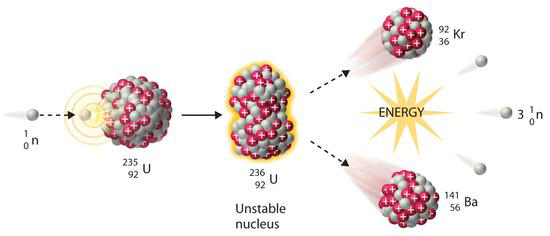 Figure XV.5. Induced fission of a heavy nucleus into two lighter nuclei
(http://chemwiki.ucdavis.edu/Physical_Chemistry/Nuclear_Chemistry/Nuclear_Reactions).
Figure XV.5. Induced fission of a heavy nucleus into two lighter nuclei
(http://chemwiki.ucdavis.edu/Physical_Chemistry/Nuclear_Chemistry/Nuclear_Reactions).
Despite fission usually being caused by a neutron, it can be produced by other particles, such as protons, deuterons and alpha particles (and even by the gamma rays) that have enough energy to cross the Coulomb barrier and introduce enough excitation energy via their kinetic energy. The requisite excitation energy of the intermediate nucleus is 4-6 MeV. While spontaneous fission only pertains to heavy nuclei, induced fission can also be achieved with medium heavy nuclei, even lanthanides, as long as the projectile particle energy is high enough, at least 50 MeV.
 Figure XV.6. Heavy nucleus fission after neutron capture.
Figure XV.6. Heavy nucleus fission after neutron capture.
The fission process can be explained as follows. Two opposing forces exist in the nucleus. The short-range nuclear forces which are smallest when the nucleus is spherical. While the longer range electrical forces, i.e. proton repulsion, aims to tear apart and deform the nucleus. Therefore, this is why, for example, the uranium nucleus is slightly elliptical, not perfectly spherical. When a neutron absorbs into the uranium nucleus it causes an increase in the ellipticity and disintegration of the nucleus if the excitation energy is sufficiently large (Figure XV.5).
Fission releases a large amount of energy, because medium heavy nuclei have a stronger binding energy to nucleons, about 8 MeV/nucleon, than heavy nuclei in which it is about 7 MeV/nucleon (see Figure II.2). For each nucleon an energy of almost 1 MeV is released and in an individual fission event of a heavy nucleus a total of 200 MeV of energy is released, the distribution of which is shown in Table XV.I
Table XV.1. The distribution of the 200 MeV total energy generated by a 235U fission event.
| Kinetic energy of fission products | 165 MeV | |
|---|---|---|
| Kinetic energy of neutrons | 5 MeV | |
| Gamma energy released immediately at fission | 7 MeV | |
| Beta particle energy of fission products | 7 MeV | |
| Gamma ray energy of fission products | 6 MeV | |
| Energy generated by neutrinos in beta decay | 10 MeV | |
In conventional fission types the fission products generated are mostly of a different size (asymmetric fission). Figure XV.7a shows the distribution of fission products of the thermal neutron induced fission of three nuclides 235U, 239Pu, and 241Pu. Events, in which fission products are of equal size (symmetric fission) are extremely rare, occurring in only 0.05-0.01% of the cases. Distribution of uranium fission products has two peaks, at a mass numbers of 90-100 and of 130-140. The largest fission yields at these mass numbers are about 6-7%. The fission nuclides 90Sr and 137Cs belong to this category: they constitute the major part of the activity of fission products within the next few hundred years. The half-life of both is relatively long, about 30 years. Plutonium also has a fission product peak at the same upper mass number, but the lower mass number peak is transferred to a higher range, 95-105. When going further into heavier fissioning nuclides this lower mass number peak range moves closer to the upper range and the valley between them narrows and becomes shallower, increasing the likelihood of symmetric fission. The probability of symmetric fission also increases when the projectile particle energy grows (Figure XV.7b): in the fission of 235U induced by 14 MeV neutrons already one out of a hundred results in a symmetric fission. When the neutron energy is raised to 100 MeV, the valley between the peaks disappears.
 Figure XV.7. Yields of fission products (%) as a function of their mass number: a) thermal neutron induced fission of 233U, 235U, 239Pu and 241Pu (https://en.wikipedia.org/wiki/Fission_product_yield#/media/File:ThermalFissionYield.svg) b): 235U fission with thermal and 14 MeV neutrons (http://www.tpub.com/doenuclearphys/nuclearphysics29.htm).
Figure XV.7. Yields of fission products (%) as a function of their mass number: a) thermal neutron induced fission of 233U, 235U, 239Pu and 241Pu (https://en.wikipedia.org/wiki/Fission_product_yield#/media/File:ThermalFissionYield.svg) b): 235U fission with thermal and 14 MeV neutrons (http://www.tpub.com/doenuclearphys/nuclearphysics29.htm).
In a fission event 2-3 neutrons, prompt neutrons, form at disintegration moment. The daughter nuclides formed in fission are always radioactive, because fissioning heavy elements have a greater neutron to proton ratio than lighter elements. Thus, the fissioning nuclides, even after emitting 2-3 neutrons, have too many neutrons and they decay via β-decay to correct the unstable neutron/proton ratio. Decay occurs in several stages forming a decay chain. The neutron to proton ratio of 235U is 1.55 and is roughly the same with the primary fission product nuclides. For example, the stable barium isotopes, however, have a much lower neutron to proton ratio of 1.32-1.46. An example of this type of beta decay chain in which the neutron/proton ratio decreases is:
$${}^{137}_{\phantom{1}52}\mathrm{Te} \rightarrow 3.5\mathrm{s} \rightarrow {}^{137}_{\phantom{1}53}\mathrm{I} \rightarrow 24.5\mathrm{s} \rightarrow {}^{137}_{\phantom{1}54}\mathrm{Xe} \rightarrow 3.8\ \mathrm{min} \rightarrow {}^{137}_{\phantom{1}55}\mathrm{Cs} \rightarrow 30\alpha \rightarrow {}^{137}_{\phantom{1}56}\mathrm{Ba} \ (\text{stable})$$
in which the n/p ratio decreases correspondingly as:
$$\text{n/p: } {}^{137}_{\phantom{1}52}\mathrm{Te} \ (1.63) \rightarrow {}^{137}_{\phantom{1}53}\mathrm{I} \ (1.58) \rightarrow {}^{137}_{\phantom{1}54}\mathrm{Xe} \ (1.54) \rightarrow {}^{137}_{\phantom{1}55}\mathrm{Cs} \ (1.49) \rightarrow {}^{137}_{\phantom{1}56}\mathrm{Ba} \ (1.45)$$
As shown, when going towards stable nuclides from the primary fission nuclides the half-lives lengthen, reflecting the increase in stability. In some beta decay events, neutrons, called delayed neutrons, are also emitted. They are only a small fraction of the prompt neutrons, e.g. 0.02% in 235U fission.
The nuclides, in which a fission reaction is possible, are called fissionable, i.e. eligible for fission. Nuclides able to undergo fission induced by thermal neutron are called fissile. Of these the most important are 235U and 239Pu, which play an important role as nuclear reactor fuel and nuclear weapons material. Of these, 235U is the only naturally occurring fissile material. Neutron bombardment of 238U produce 239Pu by neutron capture and beta decay. A characteristics of uranium, and plutonium, isotopes is that the isotopes(233U, 235U), with an odd mass number are fissile, but ones with an even mass number (234U, 238U) only undergo fission induced by high energy neutrons. This is because when all the protons in the uranium nucleus (Z = 92) are paired, the uranium isotopes with an odd mass number have unpaired neutrons. Binding energy released in the pair formation of the absorbed neutron with the unpaired neutron is enough to induce fission. Instead, with uranium isotopes with an even mass number, not enough binding energy is released to induce fission since the absorbed neutron remains unpaired. In this case, to induce fission, kinetic energy of the fast neutrons is needed. Cross sections of induced fission of 235U and 238U are seen in Figure XV.8.
Figure XV.8. Cross section of neutron induced fission of 235U and 238U (D.T.Hughes and R.B. Schwartz, Neutron Cross Sections, Brookhaven National Laboratory Report 325, 2nd Edition, 1958).
In order for fission events to continue spontaneously, become a chain reaction, in fissile material there has to be a sufficient amount of material. If there is too little material the neutrons escape without causing new fission. At least one neutron generated by a fission event must cause at least one new fission in order to create a chain reaction. The chain reaction is controlled, if only one neutron causes one new fission. If more than one neutron induces fission, it is uncontrolled fission, i.e. a bomb. The minimum mass of a spherical fissile material at which fission chain reaction occurs is called the critical mass. It is 52 kg for 235U and only 16 kg for 239Pu.
textbook/nrctextbook/chapter15.txt · Last modified: 2025-05-05 11:10 by Merja Herzig
A-CINCH Consortium
email: mst@evalion.cz | tel: +420 224 358 331 | Copyright © 2021 A-CINCH
This project has received funding from the Euratom research and training programme 2019–2020 under grant agreement No. 945301.



