User Tools
Sidebar
textbook:nrctextbook:chapter18
Table of Contents
18. Calculation Exercises
(produced by Dr. Jukka Kuva)
18.1. Activity and uncertainty calculations
18.1.1. Exercises
1. a) 0.0118% of natural potassium is radioactive 40K isotope the half-life of which is 1.27×109 years. What is the 40K activity in a human body in which the amount of potassium is 125 g? Molar mass of potassium is 39.1 g/mol.
b) What is the uncertainty of the 40K activity calculated above when the uncertainty of the potassium mass (125 g) is 15%? Use the uncertainty propagation law.
2. a) The activity of a radionuclide decreased to one per mil of the initial value in seven days. What is the half-life of the radionuclide?
b) What is the activity of 8 MBq of 90Sr after 42 years? Half-life is 28.8 years.
3. What is the 14C/C ratio in a carbon sample the specific activity of which is 50.1 dpm/g? Half-life of 14C is 5730 years.
4. How many impulses should be recorded from a radioactive sample to observe a of the number of impulses lower than 0.5%?
5. The counting efficiency of a whole-body counting system is 1.35% at 935 keV gamma energy. The background count rate at this energy was observed to be 5798 pulses in 60 minutes. A person contaminated with 115Cd was measured for 15 minutes and the number of pulses at 935 keV was 3987. What was the 115Cd activity in the body of this person? 115Cd emits gamma rays of 935 keV energy with 1.90% intensity.
6. Calculate the uncertainty of the human body activity determined in exercise 5. Use uncertainty propagation law. You do not need to take into account uncertainties of intensity, counting efficiency and counting time. Express the result as the activity with its uncertainty.
7. Marie and Pierre Curie used 2 tons of pitchblende (75% U3O8) as a starting material to isolate radium. What was the amount of 226Ra (mg) in radioactive equilibrium with the amount of uranium initially present. For simplicity we assume that all uranium was only 238U the molar mass of which is 238.03 g/mol and its half-life is 4.5×109 years. The half-life of 226Ra is 1600 years.
8. In the decay chain 238U the end-member is the stable 206Pb. From a rock sample found in Australia the amount 206Pb for each gram of 238U was 0.236 g. What was the age of the rock assuming that it did not contain any lead when it is formed?
18.1.2. Solutions
1a. 40K activity in the body:
$$A = \frac{\ln 2 \times m \times N_A}{t_{1/2} \times M} = \frac{\ln 2 \times 0.000118 \times 125\, \text{g} \times 6.022 \times 10^{23}\, \text{mol}^{-1}}{1.27 \times 10^9\, \text{a} \times 3.15 \times 10^7\, \text{s/a} \times 39.1\, \text{g/mol}} \approx 3.9\, \text{kBq}$$
1b. The uncertainty of the 40K activity:
\[\delta A = \sqrt{ \left( \frac{\partial A}{\partial m} \cdot \delta m \right)^2 } = \left| \frac{\ln 2 \cdot N_A}{t_{1/2} \cdot M} \cdot \delta m \right|\]
\[= \frac{\ln 2 \cdot 6.022 \cdot 10^{23}\, \text{mol}^{-1}}{1.27 \cdot 10^9\, \text{a} \cdot 3.15 \cdot 10^7\, \text{s/a} \cdot 39.1\, \text{g/mol}} \cdot 0.15 \cdot 0.000118 \cdot 125\, \text{g} \approx 590\, \text{Bq}\]
$$\rightarrow \delta A = 0.6\, \text{kBq}$$
2a. The half-life of the unknown radionuclide:
$$\frac{A}{A_0} = 1\,\text{‰} = 0.001$$
$$A = A_0 \cdot 2^{-\frac{t}{t_{1/2}}} \rightarrow \ln \frac{A}{A_0} = -\frac{t \cdot \ln 2}{t_{1/2}} \rightarrow t_{1/2} = -\frac{t \cdot \ln 2}{\ln \left( \frac{A}{A_0} \right)} = -\frac{7\, \text{d} \cdot \ln 2}{\ln 0.001} = 0.702\, \text{d} = 16.8\, \text{h}$$
Based on the observed half-life the nuclide can be identified to 125Xe.
2b. 90Sr activity:
$$A = A_0 \cdot 2^{-\frac{t}{t_{1/2}}} = 8\, \text{MBq} \cdot 2^{-\frac{42\, \text{a}}{28.8\, \text{a}}} \approx 2.91\, \text{MBq}$$
3. 14C/C ratio:
$$A_s^{14} = \frac{\ln 2 \cdot N_A \cdot \rho}{t_{1/2} \cdot M_C} \rightarrow \rho = \frac{A_s^{14} \cdot t_{1/2} \cdot M_C}{\ln 2 \cdot N_A} = \frac{\left(\frac{50.1}{60}\, \frac{1}{\text{s} \cdot \text{g}} \cdot 5730\, \text{a} \cdot 3.15 \cdot 10^7\, \text{s/a} \cdot 12\, \text{g/mol}\right)}{\ln 2 \cdot 6.022 \cdot 10^{23}\, \frac{1}{\text{mol}}} \approx 4.33 \cdot 10^{-12}$$
4. Number of impulses required:
$$\sigma = \sqrt{x} \leq 0.005x \rightarrow \sqrt{x} \geq \frac{1}{0.005} \rightarrow x \geq \frac{1}{0.000025} = 40\,000$$
5. 115Cd activity in the body of the person:
$$R_n = A \epsilon x = R_b - R_t \rightarrow A = \frac{R_b - R_t}{\epsilon x} = \frac{\left(\frac{3987}{15}\, \frac{1}{\text{min}} \cdot \frac{1}{60}\, \frac{\text{min}}{\text{s}} - \frac{5798}{60}\, \frac{1}{\text{min}} \cdot \frac{1}{60}\, \frac{\text{min}}{\text{s}}\right)}{0.0135 \cdot 0.019} \approx 10.99\, \text{kBq}$$
6.
$$\delta A = \sqrt{ \left( \frac{\partial A}{\partial R_b} \cdot \delta R_b \right)^2 + \left( \frac{\partial A}{\partial R_t} \cdot \delta R_t \right)^2 } = \sqrt{ \left( -\frac{\delta R_b}{\epsilon x} \right)^2 + \left( \frac{\delta R_t}{\epsilon x} \right)^2 }$$
$$= \sqrt{ \left( -\frac{\sqrt{5798/60} \, \frac{1}{\text{min}} \cdot \frac{1}{60} \, \frac{\text{min}}{\text{s}}}{0.0135 \cdot 0.019} \right)^2 + \left( \frac{\sqrt{3987/15} \, \frac{1}{\text{min}} \cdot \frac{1}{60} \, \frac{\text{min}}{\text{s}}}{0.0135 \cdot 0.019} \right)^2 } \approx 286 \, \text{Bq} \approx 300 \, \text{Bq}$$
$$\rightarrow A = (11.0 \pm 0.3) \, \text{kBq}$$
7. The amount of 226Ra:
In radioactive equilibrium: ARa = AU. m(U3O8) = 0.75×m(pitchblende) = 1.5 tons M(U3O8) =3×238.03 g/mol + 8×16.0 g/mol = 842.09 g/mol.
$$A_{238U} = \frac{\ln 2 \cdot m \cdot \rho \cdot X \cdot N_A}{t_{1/2} \cdot M} = \frac{\ln 2 \cdot 1.5 \cdot 10^6\, \text{g} \cdot 1 \cdot 3 \cdot 6.022 \cdot 10^{23}\, \frac{1}{\text{mol}}}{4.5 \cdot 10^9\, \text{a} \cdot 3.1536 \cdot 10^7\, \frac{\text{s}}{\text{a}} \cdot 842.09\, \text{g/mol}} \approx 1.571849 \cdot 10^{10}\, \text{Bq}$$
$$m_{226Ra} = \frac{A_{226Ra} \cdot M \cdot t_{1/2}}{\ln 2 \cdot N_A} = \frac{1.5718 \cdot 10^{10}\, \text{Bq} \cdot 226\, \text{g/mol} \cdot 1600\, \text{a} \cdot 3.1536 \cdot 10^7\, \text{s/a}}{\ln 2 \cdot 6.022 \cdot 10^{23}\, \frac{1}{\text{mol}}} \approx 0.4294\, \text{g} \approx 430\, \text{mg}$$
8. All lead is generated from U, thus the age of the rock is
$$N_{Pb} = N_{0U} - N_U \rightarrow N_U = N_{0U} - N_{Pb} = N_{0U} \cdot 2^{-\frac{t}{t_{1/2}}} \rightarrow \frac{N_U}{N_U + N_{Pb}} = 2^{-\frac{t}{t_{1/2}}} \rightarrow \ln \left( \frac{N_U}{N_U + N_{Pb}} \right) = -\frac{t}{t_{1/2}} \cdot \ln 2$$
$$\rightarrow t = -\frac{t_{1/2} \cdot \ln\left(\frac{N_U}{N_U + N_{Pb}}\right)}{\ln 2}$$
$$= -\frac{t_{1/2} \cdot \ln\left(\frac{\frac{m_U}{M_U} \cdot N_A}{\frac{m_U}{M_U} \cdot N_A + \frac{m_{Pb}}{M_{Pb}} \cdot N_A}\right)}{\ln 2}$$
$$= -\frac{4.47 \cdot 10^9 \, a \cdot \ln\left(\frac{\frac{1 \, g}{238 \, g/mol}}{\frac{1 \, g}{238 \, g/mol} + \frac{0.236 \, g}{206 \, g/mol}}\right)}{\ln 2} \approx 1.55 \, Ga$$
18.2. Count rate and dead-time
Chapter 8
18.2.1. Exercises
1. A sample with an activity of 55.0 kBq was measured with a GM counter and number of observed net counts was 280 000. Another sample with an activity of 55.0 Bq, obtained by diluting the first sample by a factor of 1000, was then measured with the same GM counter in identical conditions. In this case the number of pulses was 350. Calculate the counting efficiency of the counting system and dead-time of the GM detector.
2. Calculate the uncertainties of counting efficiency and dead-time observed in exercise 1, using uncertainty propagation law, for a case where the uncertainty of both samples was 2%. Express the results as the counting efficiency and the dead-time with their uncertainties.
3. An unknown radionuclide was measured with a GM counter having a dead-time of 32 µs. The observed net count rate was 280 000 imp/min on March 15th and 123 imp/min on November 10th of the same year. We assume the counting efficiency having been constant. Calculate the half-life of the radionuclide.
18.2.2. Solutions
1.
At the lower count rate the dead-time has no effect and the counting efficiency can be calculated as:
$$ R = Ax\epsilon \implies \epsilon = \frac{R_2}{A_2 x} = \frac{350 \, \text{min}^{-1} \cdot \frac{1}{60} \, \text{min/s}}{55 \, \text{s}^{-1} \cdot 1} \approx 10.6\% $$
The dead-time is calculated as:
$$R_{01} = A_1 \cdot \epsilon = 55000 \, \text{s}^{-1} \cdot 0.1060606\ldots \approx 5833.33\ldots \, \text{s}^{-1}$$
$$ R_{01} = \frac{R_1}{1 - R_1 \tau} \implies \tau = \frac{R_{01} - R_1}{R_{01} \cdot R_1} = \frac{5833 \, \text{s}^{-1} - 280000 \, \text{min}^{-1} \cdot \frac{1}{60} \, \text{min/s}}{5833 \, \text{s}^{-1} \cdot 280000 \, \text{min}^{-1} \cdot \frac{1}{60} \, \text{min/s}} \approx 4.286 \cdot 10^{-5} \, \text{s} \approx 42.9 \, \mu\text{s}$$
2. The uncertainty of the counting efficiency:
$$\delta \epsilon = \sqrt{\left( \frac{\partial \epsilon}{\partial A} \cdot \delta A \right)^2 + \left( \frac{\partial \epsilon}{\partial R} \cdot \delta R \right)^2} = \sqrt{\left( -\frac{R}{A^2 x} \cdot \delta A \right)^2 + \left( \frac{\delta R}{A x} \right)^2}$$
$$\sqrt{\left( -\frac{350 \, \text{min}^{-1} \cdot \frac{1}{60} \, \text{min/s}}{(55 \, \text{s}^{-1})^2 \cdot 1} \cdot 0.02 \cdot 55 \, \text{s}^{-1} \right)^2 + \left( \frac{\sqrt{350 \, \text{min}^{-1} \cdot \frac{1}{60} \, \text{min/s}}}{55 \, \text{s}^{-1} \cdot 1} \right)^2} \approx 0.00605 \approx 0.7\%$$
$$\implies \epsilon = (10.6 \pm 0.7)\%$$
The uncertainty of the dead-time:
$$\tau = \frac{A \epsilon - R}{A \epsilon \cdot R} = \frac{1}{R} - \frac{1}{A \epsilon}$$
$$\delta \tau = \sqrt{\left( \frac{\partial \tau}{\partial A} \cdot \delta A \right)^2 + \left( \frac{\partial \tau}{\partial R} \cdot \delta R \right)^2 + \left( \frac{\partial \tau}{\partial \epsilon} \cdot \delta \epsilon \right)^2}$$
$$\sqrt{\left( \frac{\delta A}{A^2 \epsilon} \right)^2 + \left( -\frac{\delta R}{R^2} \right)^2 + \left( \frac{\delta \epsilon}{A \epsilon^2} \right)^2}$$
$$\sqrt{\left( \frac{0.05 \cdot 55000 \, \text{s}^{-1}}{(55000 \, \text{s}^{-1})^2 \cdot 0.106} \right)^2 + \left( -\frac{\left( \sqrt{280000} \, \text{min}^{-1} \cdot \frac{1}{60} \, \text{min/s} \right)^2}{(280000 \, \text{min}^{-1} \cdot \frac{1}{60} \, \text{min/s})^2} \right)^2 + \left( \frac{0.008}{55000 \, \text{s}^{-1} \cdot (0.106)^2} \right)^2}$$
$$\approx 13.39 \, \mu\text{s} \approx 14 \, \mu\text{s}$$
$$\tau = (43 \pm 14) \, \mu\text{s}$$
3. Dead-time correction is needed only for the higher count rate. Below 1 refers to March and 2 to November.
$$A_2 = A_1 \cdot 2^{-\frac{t}{t_{1/2}}} \implies t_{1/2} = -\frac{\ln 2 \cdot t}{\ln \left( \frac{A_2}{A_1} \right)} = -\frac{\ln 2 \cdot t}{\ln \left( \frac{R_{02}/\epsilon}{R_{01}/\epsilon} \right)} = -\frac{\ln 2 \cdot t}{\ln \left( \frac{R_2}{R_1/(1 - R_1 \tau)} \right)}$$
$$= -\frac{\ln 2 \cdot 240 \, \text{d}}{\ln \left( \frac{\frac{123}{60} \, \text{s}^{-1}}{\frac{280000}{60} \, \text{s}^{-1}} \bigg/ \left(1 - \frac{280000}{60} \, \text{s}^{-1} \cdot 32 \cdot 10^{-6} \, \text{s}\right) \right)} = 21.0787\ldots \, \text{d} \approx 21 \, \text{d}$$
18.3. Nuclear reactions, production of radionuclides, succesive radioactive decay
18.3.1. Exercises
1. Molybdenum was irradiated with thermal neutrons to produce radioactive 99Mo (t1/2 = 2.75 d) isotope from stable 98Mo isotope. 99Mo decays by beta decay to 99Tc (t1/2 = 2.13 x 105 a) which further decays by beta decay to stable 99Ru. After irradiation molybdenum was separated from the target material; the 99Mo activity in the separated molybdenum was 100 kBq. What were the 99Mo and 99Tc activities after a) 1 h, b) 3d, c) 1a ? Use equation: $$A_2 = \frac{\lambda_2 \lambda_1}{\lambda_2 - \lambda_1} N_1^0 \left( 2^{-\frac{t}{t_1}} - 2^{-\frac{t}{t_2}} \right)$$
2. A fish sample of 2.0 g weight was irradiated in a reactor for 2.0 hours with a neutron flux of 5.0×1017 (φ). Twenty days after the irradiation the activity of generated 203Hg was measured to be 226 Bq. Calculate the mercury concentration of the fish as mg/kg. The cross section (σ) of the reaction 202Hg(n,γ)203Hg is 380 fm2, the half-life of 203Hg is 47 days, the isotopic abundance of 202Hg is 29.8% and the atomic weight of mercury is 200.6 . Use equation:
$$A_\tau = \sigma \phi N_1 \left( 1 - 1^{-\frac{t_S}{T}} \right) 2^{-\frac{t}{T}}$$
3. To determine the neutron flux of a reactor, 10 mg of copper was irradiated for 4.0 hours. 25 hours and 36 minutes after the irradiation was terminated the 64Cu activity was 70 MBq. The cross section of the reaction 63Cu(n,γ)64Cu is 4.51 barn, the isotopic abundance of 63Cu is 69.1%, the atomic weight of copper is 63.5 g/mol and the half-life of 64Cu is 12.8 hours. What is the neutron flux of the reactor?
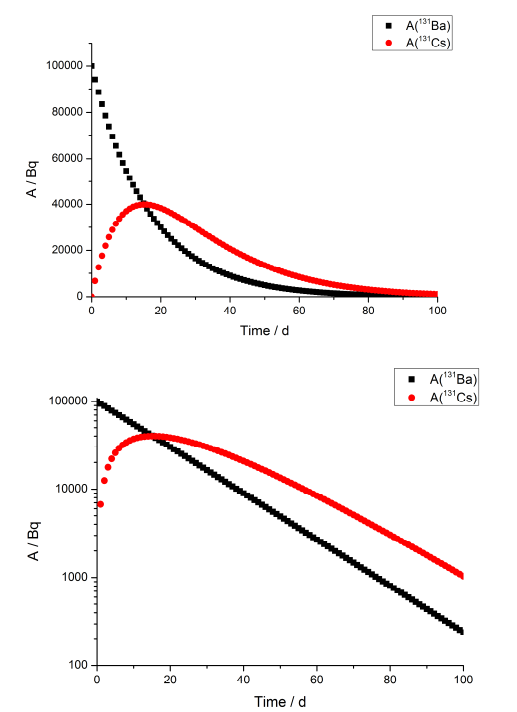
4. 131Ba (t1/2 = 11.5 d) decays by positron emission to 131Cs (t1/2 = 9.7 d) and the latter further to stable 131Xe. Barium is separated from a sample containing both nuclides. What are the 131Ba and 131Cs activities in the separated barium fraction after 1, 10 and 100 days when the initial 131Ba activity was 100 kBq. Plot, with excel or other suitable program, the activities of both nuclides for the time period up to 100 days. When the nuclide activities are identical? When the 131Cs activity has its maximum?
18.3.2. Solutions
1. Calculation of decay constants:
$$A_1^0 = 100 \, \text{kBq}$$ $$\lambda_1 = \frac{\ln 2}{T_1} = \frac{\ln 2}{2.75 \cdot 24 \cdot 3600 \, \text{s}} \approx 2.917 \cdot 10^{-6} \, \text{s}^{-1}$$ $$\lambda_2 = \frac{\ln 2}{T_2} = \frac{\ln 2}{2.13 \cdot 10^5 \cdot 365 \cdot 24 \cdot 3600 \, \text{s}} \approx 1.032 \cdot 10^{-13} \, \text{s}^{-1}$$ $$N_1^0 = \frac{A}{\lambda_1} = \frac{1 \cdot 10^5 \, \text{1/s}}{2.917 \cdot 10^{-6} \, \text{1/s}} \approx 3.43 \cdot 10^{10}$$
Inserting these and the half-lives to the equation
$$A_2 = \frac{\lambda_2 \lambda_1}{\lambda_2 - \lambda_1} N_1^0 \left(2^{-\frac{t}{t_1}} - 2^{-\frac{t}{t_2}}\right)$$
yields the following activities for the three time points:
| t | A1(99Mo) | 2-t/T1 | 2-t/T2 | A2(99Tc) |
|---|---|---|---|---|
| 1 h | 99.0 kBq | 0.989 | 1 | 36.9 μBq |
| 3 d | 46.9 kBq | 0.469 | 1 | 1.8 mBq |
| 1 a | 0 kBq | 0 | 1 | 3.5 mBq |
2.
$$A_\tau = \sigma \phi N_1 \left(1 - 1^{-\frac{t_S}{T}}\right) 2^{-\frac{t}{T}}$$
and
$$N_1 = \frac{m \cdot \rho_i \cdot N_A}{M}$$
⇒
$$\rho = \frac{m_{Hg}}{m} = \frac{N_1 M}{m \rho_i N_A} = \frac{A_t M}{\sigma \cdot \phi \cdot \left(1 - 2^{-\frac{t_S}{T}}\right) \cdot 2^{-\frac{t}{T}} \cdot m \cdot \rho_i \cdot N_A}$$
$$= \frac{226 \, \text{Bq} \cdot 200.6 \, \text{g/mol}}{380 \cdot 10^{-30} \, \text{m}^2 \cdot 5 \cdot 10^{17} \, \text{1/(m}^2 \text{s)} \cdot \left(1 - 2^{-\frac{2h}{47d \cdot 24 \, \text{h/d}}}\right) \cdot 2^{-\frac{20d}{47d}} \cdot 2 \, \text{g} \cdot 0.298 \cdot 6.022 \cdot 10^{23} \, \text{1/mol}}$$
$$\approx 7.2697 \cdot 10^{-7} \, \text{g/g} \approx 0.73 \, \text{mg/kg}$$
3.
$$\phi = \frac{A_t}{\sigma N_1 \left(1 - 2^{-\frac{t_S}{T}}\right) 2^{-\frac{t}{T}}} = \frac{A_t M}{\sigma m N_A \rho \left(1 - 2^{-\frac{t_S}{T}}\right) 2^{-\frac{t}{T}}}$$
$$= \frac{70 \cdot 10^6 \, \text{Bq} \cdot 63.5 \, \text{g/mol}}{4.51 \cdot 10^{-28} \, \text{m}^2 \cdot 0.01 \, \text{g} \cdot 6.022 \cdot 10^{23} \, \text{1/mol} \cdot 0.691 \cdot \left(1 - 2^{-\frac{4h}{12.8h}}\right) 2^{-\frac{25h + 36/60 \, h}{12.8h}}}$$
$$\approx 4.86 \cdot 10^{16} \, \text{1/(m}^2 \text{s)}$$
4. Calculation of decay constants:
$$A_1^0 = 100 \, \text{kBq}$$
$$\lambda_1 = \frac{\ln 2}{T_1} = \frac{\ln 2}{11.5 \cdot 24 \cdot 3600 \, \text{s}} \approx 6.97611 \cdot 10^{-7} \, \text{1/s}$$
$$\lambda_2 = \frac{\ln 2}{T_2} = \frac{\ln 2}{9.7 \cdot 24 \cdot 3600 \, \text{s}} \approx 8.27066 \cdot 10^{-7} \, \text{1/s}$$
$$N_1^0 = \frac{A}{\lambda_1} = \frac{1 \cdot 10^5 \, \text{1/s}}{6.97611 \cdot 10^{-7} \, \text{1/s}} \approx 1.43346 \cdot 10^{11}$$
Inserting these and the half-lives to the equation
$$A_2 = \frac{\lambda_2 \lambda_1}{\lambda_2 - \lambda_1} N_1^0 \left(2^{-\frac{t}{t_1}} - 2^{-\frac{t}{t_2}}\right)$$
yields the following activities for the three time points:
| t | A1(131Ba) | 2-t/T1 | 2-t/T2 | A2(131Cs) |
|---|---|---|---|---|
| 1 d | 94.2 kBq | 0.942 | 0.931 | 6.69 kBq |
| 10 d | 54.7 kBq | 0.547 | 0.489 | 37.0 kBq |
| 100 d | 241 Bq | 0.00241 | 0.000788 | 1.04 kBq |
The activities are equal at time point 15 days where the maximum of 131Cs activity also is observed.
18.4. Applications to some other problems
18.4.1. Exercises
1. A medical doctor determines the blood volume of a patient by injecting 0.15 ml of 99mTcO4 (t½= 6.01 hours) solution into the patient’s blood vessel. The activity concentration of the solution was 30 MBq/mL at the time of injection. After 4 hours a sample of the blood is taken and its activity concentrations is measured to be 610 Bq/mL. Calculate the total blood volume of the patient.
2. Calculate the uncertainty of the blood volume determined in exercise 1. The uncertainty of the injection volume was 0.01 ml and the relative uncertainties of the initial and final 99mTc activities were 5% (uncertainty of the counting time is very small and is not taken into account). Calculate the uncertainty by using uncertainty propagation law and express the result as the total blood volume with its uncertainty.
3. 226Radium (M = 226.025 g/mol) decays to 222Rn with a half-life of 1600 years. What is the volume of the 222Rn gas generated from 42 kg of Ra (T=25 °C ja p = 1 atm)?
18.4.2. Solutions
1.
Activity concentration of 99mTc in blood at the time of injection:
$$A = A_0 \cdot 2^{-\frac{t}{t_{1/2}}} \rightarrow A_0 = \frac{A}{2^{-\frac{t}{t_{1/2}}}} = \frac{610 \, \text{Bq/mL}}{2^{-\frac{4h}{6.01h}}} \approx 967.5704 \, \text{Bq/mL}$$
The blood volume (assuming homogenous distribution of 99mTc in blood):
$$A_{\text{inj}} \cdot V_{\text{inj}} = A_{\text{blood}} \cdot V_{\text{blood}} \rightarrow V_{\text{blood}} = \frac{A_{\text{inj}} \cdot V_{\text{inj}}}{A_{\text{blood}}} = \frac{30 \cdot 10^6 \, \text{Bq/mL} \cdot 0.15 \, \text{mL}}{967.5704 \, \text{Bq/mL}} \approx 4.65 \, \text{L}$$
2. $$V_{\text{blood}} = \frac{A_{\text{inj}} \cdot V_{\text{inj}}}{A_{\text{blood}}} = \frac{A_{\text{inj}} \cdot V_{\text{inj}} \cdot 2^{-\frac{t}{t_{1/2}}}}{A_{\text{blood sample}}}$$
$$\delta V_{\text{blood}} = \sqrt{\left(\frac{\partial V_{\text{blood}}}{\partial A_{\text{inj}}} \cdot \delta A_{\text{inj}}\right)^2 + \left(\frac{\partial V_{\text{blood}}}{\partial A_{\text{blood sample}}} \cdot \delta A_{\text{blood sample}}\right)^2 + \left(\frac{\partial V_{\text{blood}}}{\partial V_{\text{inj}}} \cdot \delta V_{\text{inj}}\right)^2}$$
$$= \sqrt{\left(\frac{V_{\text{inj}} \cdot 2^{-\frac{t}{t_{1/2}}} \cdot \delta A_{\text{inj}}}{A_{\text{blood sample}}}\right)^2 + \left(-\frac{A_{\text{inj}} \cdot V_{\text{inj}} \cdot 2^{-\frac{t}{t_{1/2}}} \cdot \delta A_{\text{blood sample}}}{(A_{\text{blood sample}})^2}\right)^2 + \left(\frac{A_{\text{inj}} \cdot 2^{-\frac{t}{t_{1/2}}} \cdot \delta V_{\text{inj}}}{A_{\text{blood sample}}}\right)^2}$$
$$= \sqrt{ \begin{array}{l} \left(\frac{0.15 \, \text{mL} \cdot 2^{-\frac{4h}{6.01h}} \cdot 0.05 \cdot 30 \cdot 10^6 \, \text{Bq/mL}}{610 \, \text{Bq/mL}}\right)^2 + \\ \left(-\frac{30 \cdot 10^6 \, \text{Bq/mL} \cdot 0.15 \, \text{mL} \cdot 2^{-\frac{4h}{6.01h}} \cdot 0.05 \cdot 610 \, \text{Bq/mL}}{(610 \, \text{Bq/mL})^2}\right)^2 + \\ \left(\frac{30 \cdot 10^6 \, \text{Bq/mL} \cdot 2^{-\frac{4h}{6.01h}} \cdot 0.01 \, \text{mL}}{610 \, \text{Bq/mL}}\right)^2 \end{array} }$$
$$\approx 451.98 \, \text{mL} \approx 500 \, \text{mL}$$
$$\rightarrow V_{\text{blood}} = (4.7 \pm 0.5) \, \text{L}$$
3.
first the number of generated 222Rn atoms is calculated:
$$N_{\text{Rn}} = N_{Q\text{Ra}} - N_{\text{Ra}} = N_{Q\text{Ra}} - N_{Q\text{Ra}} \cdot 2^{-\frac{t}{t_{1/2}}} = \frac{m_{\text{Ra}}}{M_{\text{Ra}}} \cdot N_A \cdot \left(1 - 2^{-\frac{t}{t_{1/2}}}\right)$$
using the ideal gas law the volume can be calculated:
$$pV = nRT \rightarrow V = \frac{nRT}{N_A p} = \frac{4.846678 \cdot 10^{22} \cdot 0.08206 \, \text{(L} \cdot \text{atm)/(mol} \cdot \text{K)} \cdot 298.15 \, \text{K}}{6.022 \cdot 10^{23} \, \text{1/mol} \cdot 1 \, \text{atm}} \approx 1.97 \, \text{L}$$
textbook/nrctextbook/chapter18.txt · Last modified: 2025-05-05 13:25 by Merja Herzig
A-CINCH Consortium
email: mst@evalion.cz | tel: +420 224 358 331 | Copyright © 2021 A-CINCH
This project has received funding from the Euratom research and training programme 2019–2020 under grant agreement No. 945301.