User Tools
Sidebar
textbook:nrctextbook:chapter6
Table of Contents
6. Rate of radioactive decay
Chapter 6 from BASICS OF NUCLEAR PHYSICS AND OF RADIATION DETECTION AND MEASUREMENT – An open-access textbook for nuclear and radiochemistry students by Jukka Lehto
6.1. Decay law – activity - decay rate
The rate of radioactive decay is a characteristic feature for each radionuclide. Decay rate, also called activity, is the number of nuclear transformations (decays) (dN) at a defined time difference (unit time) (dt) and it is referred to as A (activity):
$$A = \left| -\frac{dN}{dt} \right|$$
[VI.I]
Radioactive decay is a stochastic phenomenon and we cannot know when a single nucleus will decay or what is the exact number of decays in unit time. If a fairly large number of radioactive nuclei are considered we can, however, know what fraction of nuclei will probably decay in unit time. This fraction, the probability of radioactive decay events, is characteristic for each radionuclide and it is called decay constant (λ). If, for example, the decay constant is 0.0001 s-1 it means that among 100000 radioactive nuclei probably 10 nuclei will decay in one second and accordingly among 1000000 nuclei probably 100 nuclei. Thus radioactive decay rate is directly proportional to the number of radioactive nuclei.
$$A = \left| -\frac{dN}{dt} \right| = \lambda \cdot N$$
[VI.II]
where N is the number of radioactive nuclei.
In the following we will see what is the number of radioactive nuclei (N) at defined time point (t) when their initial number (N0) is known at time point t0. From the equation VI.II we get $\frac{dN}{N} = -\lambda \cdot dt$ and its integration $\int \frac{dN}{N} = \int -\lambda \cdot dt$ yields $\ln N = -\lambda \cdot t + C$. When considering time t = 0, when N = N0 , the constant C gets a value lnN0 and inserting this into the equation $\ln N = -\lambda \cdot t + C$ yields $\ln N - \ln N_0 = -\lambda \cdot t$ and further $\ln \frac{N}{N_0} = -\lambda \cdot t$. Taking antilogarithm from both sides yields $\frac{N}{N_0} = e^{-\lambda t}$ and further
$$N = N_0 \cdot e^{-\lambda t}$$
[VI.III],
which equation answers the question what is the number of radioactive nuclei (N) at certain time point (t) when we know the initial number on nuclei (N0) at time point t0. Thus, to calculate this only the value of the decay constant (λ) is to be known.
The number of radioactive nuclei is not usually known and their number is also difficult to directly determine. Usually we are, however, more interested in development of activities with time. As seen from Equation VI.II the activity is directly proportional to the number of decaying nuclei, thus we can transform Equation VI.III to calculate activity (A) at a time point (t) just by replacing N with A:
$$A = A_0 \cdot e^{-\lambda \cdot t}$$
[VI.IV]
6.2. Half-life
Decay constants are known for all radionuclides and they are tabulated in various textbooks and databases. They are, however, not used in calculations of activities but instead half-lives (t½) are used for this purpose. Half-life is defined as the time in which half of the initial radioactive nuclei have decayed. Since the activity is directly proportional to the number of radioactive nuclei this means that also activity decreases to half within the time of half-life. In the following, the relation between the decay constant and the half-life will be shown. In addition, an equation by which activities can be calculated at desired time points using half-lives will be derived.
Do not use upper-case T½ for half-life, use lower lower-case t½. T refers to temperature, t to time.
We consider a time difference equal to a half-life $t = t_{1/2}$, during which time the number of radioactive nuclei decays to half, i.e. $N = \frac{N_0}{2}$. Inserting $t = t_{1/2}$ and $N = \frac{N_0}{2}$ to Equation VI.III $N = N_0 \cdot e^{-\lambda t}$ yields $\frac{N_0}{2} = N_0 \cdot e^{-\lambda \cdot t_{1/2}}$ and further $e^{\lambda t_{1/2}} = 2$. Taking logarithms from both sides gives $\lambda t_{1/2} = \ln 2$ and further $\lambda = \frac{\ln 2}{t_{1/2}}$. Replacing λ in equations $N = N_0 \cdot e^{-\lambda t}$, $A = A_0 \cdot e^{-\lambda t}$ with $\frac{\ln 2}{t_{1/2}}$ yields
$$N = N_0 \cdot 2^{-\frac{t}{t_{1/2}}}$$
[VI.V]
and
$$A = A_0 \cdot 2^{-\frac{t}{t_{1/2}}}$$
[VI.VI]
With the latter equation we can calculate activities using half-lives at any time points when we know the initial activity. When we want to calculate the initial activity at an earlier time point, we use the inverse equation
$$A_0 = A \cdot 2^{\frac{t}{t_{1/2}}}$$
[VI.VII]
6.3. Activity unit
The official SI unit of activity is Becquerel (Bq) and it means one decay in SI unit time, i.e. one second:
$$1 \text{ Bq} = 1 \text{ decay} \cdot \text{s}^{-1}$$
[VI.VIII]
Earlier Curie (Ci) was used as the activity unit. One Curie is 3.7×1010 decays in second and thus
$$1 \text{ Ci} = 3.7 \times 10^{10} \text{ s}^{-1} = 3.7 \times 10^{10} \text{ Bq}$$
[VI.IX]
Curie unit was derived as the number of decays taking place in one gram of 226Ra in one second using half-life of 1580 years (today it is known to be 1600 years).
Sometimes activities are expressed as a dps unit, meaning disintegrations per second which are equal to activities presented as Becquerels. In some instances, for example in liquid scintillation counting, activity is also presented as dpm units (disintegrations per minute). One dpm is 1/60 dps or 16.7 mBq.
6.4. Specific activity - activity concentration
Specific activity is often used as a synonym to activity concentration, but strictly speaking they have different meanings. Specific activity refers to concentration of a radionuclide with respect to the total amount of the same element as the radionuclide. Thus, specific activity is its concentration in a unit mass or mole of the same element, for example, 5 kBq of 137Cs per 1 g of Cs or 0.038 kBq of 137Cs per 1 mole of Cs.
Activity concentration in turn is the concentration of a radionuclide in a unit mass or volume of any matter in question, for example, 5 kBq of 137Cs per 1 kg of soil or 5 kBq of 137Cs per 1 litre of water.
6.5. The relation between the activity and the mass
Conversion of activities to masses or vice versa is based on the radioactive decay law
$$A = \lambda \times N$$
[VI.X]
which shows the direct dependence of the activity on the number of decaying nuclei. Replacing 𝜆 by $\frac{\ln 2}{t_{1/2}}$ ln2/t1/2 and N by $\left( \frac{m}{M} \right) \times N_A$ (m/M)×NA (where m is the mass in grams, M the molar mass of the element and NA the Avogadro number) yields
$$A = \frac{\ln 2 \times m \times N_A}{t_{1/2} \times M}$$
[VI.XI]
or the other way round
$$m = \frac{A \times M \times t_{1/2}}{\ln 2 \times N_A}$$
[VI.XII]
which can be used to convert activities to masses or vice versa.
6.6. Determination of half-lives
The determination of half-lives can be accomplished in two ways:
- For radionuclides decaying with such a fast rate that we can observe the decrease in a reasonable time the half-lives can be determined from their activities as a function of time as shown below in Figure VI.1.
When representing graphically the equation $A = A_0 \cdot 2^{-\frac{t}{t_{1/2}}}$ we get an exponential curve (Figure VI.1,left side). Taking logarithms from both sides yields the equation $\ln A = -\frac{\ln 2}{t_{1/2}} \cdot t + \ln A_0$, the graphical representation of which is line with a slope of $-\frac{\ln 2}{t_{1/2}}$ and the y-axis intersection is $\ln A_0$, i.e. activity at time t0 (Figure VI.1, right side). The half-life is obtained by fitting a line to the logarithms of observed activity values and calculating the half-life from the slope. If, for example, in Figure VI.1 time were in years, the half-life of the nuclide would result by solving the equation $-0.693 = -\frac{\ln 2}{t_{1/2}}$ into 1 year.
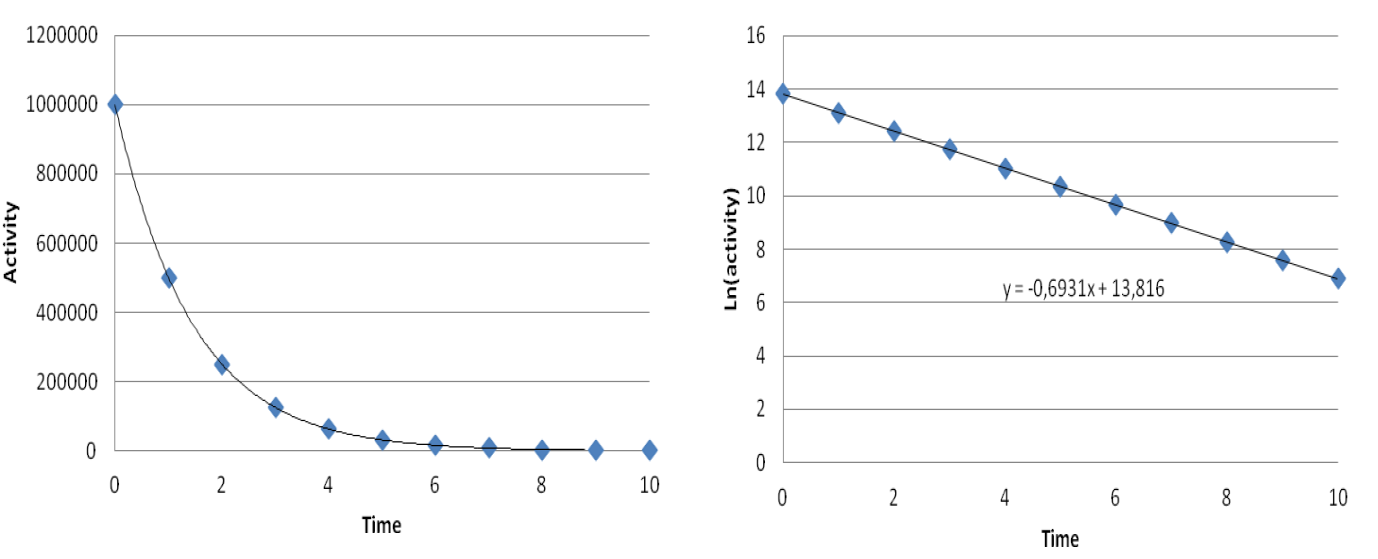 Figure VI.1. Activity (A) as a function of time (t). Left side presents activity in linear scale and right side in logarithmic scale.
Figure VI.1. Activity (A) as a function of time (t). Left side presents activity in linear scale and right side in logarithmic scale.
The method described above in Figure VI.1 can also be used to determine half-lives of two coexisting radionuclides supposing that they differ enough from each other. Figure VI.2 shows the total activity curve of two radionuclides as a function of time both in a linear and a logarithmic activity scale. In the first phase, when there are still both radionuclides present, the logarithmic curve shape resembles an exponential one. As the shorter-lived radionuclide has decayed the curve turns into a line. This line represents the decay of the longer-lived radionuclide and its half-life can be calculated from the slope of this line. To calculate the half-life of the shorter-lived radionuclide the line is extrapolated to time point zero and the extrapolated activity values of the line are subtracted from total activity curve. This yields another line representing the decay of the shorter-lived radionuclide for which the half-life is calculated from its slope.
Figure VI.2. Individual and total activities of two coexisting radionuclides as a function of time. Top: activity in linear scale. Bottom: activity in logarithmic scale. Diamond (blue): Radionuclide with a half-life of 3 hours. Square (red): radionuclide with a half-life of 12 hours. Triangle (green): total activity.
- For long-lived radionuclides for which the activity decreases so slowly that we cannot observe its decrease the half-life is determined by measuring both the activity and the mass of the radionuclide and calculating half-life from the equation $t_{1/2} = \frac{\ln 2 \times m \times N_A}{A \times M}$.
For example, when the half-life is 106 years the activity decreases by 0.00007% in one year. Small differences in activity like this cannot be measured. To determine the half-life of long-lived radionuclides we need to measure the mass (m) of the radionuclide and count rate (R) obtained from the activity measurement. In addition, we also need to accurately know the counting efficiency (E) of the measurement system. If, for example, we have 1.27 mg of 232Th and the count rate obtained from its measurement is 2.65 cps and counting efficiency of the measurement system is 51.5% (0.515) the activity of the sample is $A = \frac{R}{E} = \frac{2.65 \, \text{s}^{-1}}{0.515} = 5.15 \, \text{Bq}$. The number of thorium atoms in the sample is $1.27 \cdot 10^{-3} \, \text{g} \times \frac{6.023 \cdot 10^{23} \, \text{atoms/mole}}{232.0 \, \text{g/mole}} = 3.295 \cdot 10^{18}$. Now the half-life can be calculated from the equation $t_{1/2} = \left( \frac{\ln 2}{A} \right) \times N = \frac{0.693 \times 3.295 \cdot 10^{18}}{5.15 \, \text{s}^{-1}} = 4.44 \cdot 10^{17} \, \text{s} = 1.41 \cdot 10^{10} \, \text{a}$. This method can also in principle be used to measure half-lives of shorter lived radionuclides but in their case the accurate measurement of the mass may either completely exclude the use of this method or at least results in inaccurate value.
6.7. Activity equilibria of consecutive decays
In the following we discuss two consecutive decay processes and their activity equilibria. Equilibrium means that the activities of the parent and daughter nuclides are the same. Consecutive decays and their equilibria are especially important in natural decay chains of uranium and thorium and in beta decay chains following fissions. In these chains there are typically more than two radionuclides present at the same time. Their equilibrium calculations are rather complicated and require computer programs. An example of such calculation is given in Figure VI.7 (at the end of this chapter). Here we, however, focus on equilibrium between two radionuclides, the parent nuclideItalic Text and the daughter nuclide. When considering two consecutive decays the number of the parent nuclei (N1) depends only on its characteristic decay rate, i.e. decay constant (λ1). The number of the daughter nuclei (N2) in turn is dependent both on its own decay rate (λ2) and on the parent's decay rate (λ1). The former determines the decay (decrease) of the daughter nuclides while the latter determines the ingrowth from the parent (increase). Thus the number of daughter nuclei is
$$\frac{dN_2}{dt} = \lambda_1 N_1 - \lambda_2 N_2$$
[VI.XIII]
The solution of this equation with respect to N2 is
$$N_2 = \left[ \frac{\lambda_1}{\lambda_2 - \lambda_1} \right] N_1^0 \left( e^{-\lambda_1 t} - e^{-\lambda_2 t} \right) + N_2^0 e^{-\lambda_2 t}$$
[VI.XIV]
where N10 and N20 are the numbers of parent and daughter, respectively, at time point zero (t=0). The first term in the equation presents the number of daughter nuclei due to ingrowth and the decay of ingrown nuclei while the second term represents decay of those daughter nuclei that were present at time point zero. Figure VI.3 gives a graphical presentation of the Eq. VI.XIV for a case where the half-life of the daughter is clearly shorter than that of the parent nuclide, i.e. its shows the ingrowth of the daughter nuclide activity as a function of the number half-lives of the daughter nuclide. Activity is here presented as the percentage of the maximum activity obtainable.
Figure VI.3. Activity (percentage of the maximum activity) of the daughter nuclide as a function of the number of daughter nuclide’s half-life. The half-life of the daughter is clearly shorter than that of the parent nuclide. No daughter nuclides present at time 0.
In the following we will examine how the equilibrium develops in cases where we have initially only the parent nuclide and daughter nuclide grows in with time. There are three alternatives depending on the ratio of the half-lives of the two nuclides:
- Secular equilibrium, in which the half-life of the parent nuclide is very long and the half-life of the daughter nuclide is considerably shorter than that of the parent
- Trancient equilibrium, in which the half-life of the parent is so short that we observe decrease in its activity in a reasonable time and the half-life of the daughter nuclide is shorter than that of the parent
- Non-equilibrium, in which the half-life of the parent nuclide is shorter than that of the daughter
6.7.1. Secular equilibrium
An example of a secular equilibrium is case where the parent nuclide is the fission product 137Cs which decays by beta decay process to 137mBa which in turn decays by internal transition process to stable 137Ba. The half-life of 137Cs is 30 years while the half-life of 137Ba is only 2.6 minutes. Figure VI.3 shows the development of activities in a case when 137mBa has been chemically separated from its parent 137Cs with BaSO4 precipitation and both 137mBa-bearing precipitate and remaining solution containing only 137Cs are measured for their 137Cs and 137mBa activities immediately after chemical separation and measurements are repeated as a function of time. 137mBa in precipitate decays following its half-life on 2.6 minutes (red). The activity of 137Cs in the solution phase (blue) remains practically constant since observation time (30 min) is extremely short compared to the half-life of 137Cs (30 years). 137mBa in the solution (green) starts immediately after chemical separation to grow in and attains equilibrium with 137Cs in about ten half-lives of the daughter, i.e. half an hour. Since the activities of 137mBa and 137Cs are the same the total activity (curve 4) is twice the parent nuclide.
Secular does not mean eternal. Looking at a very long-term all secular equilibria are transient. How long-term we need to look depends on the half-life of the parent. For example, if we looked the example describe above for a hundred years period the equilibrium would appear as transient equilibrium. For 230Th (t1/2 = 75000 y), for example, the transient equilibrium period with 226Ra would be hundreds of thousands of years.
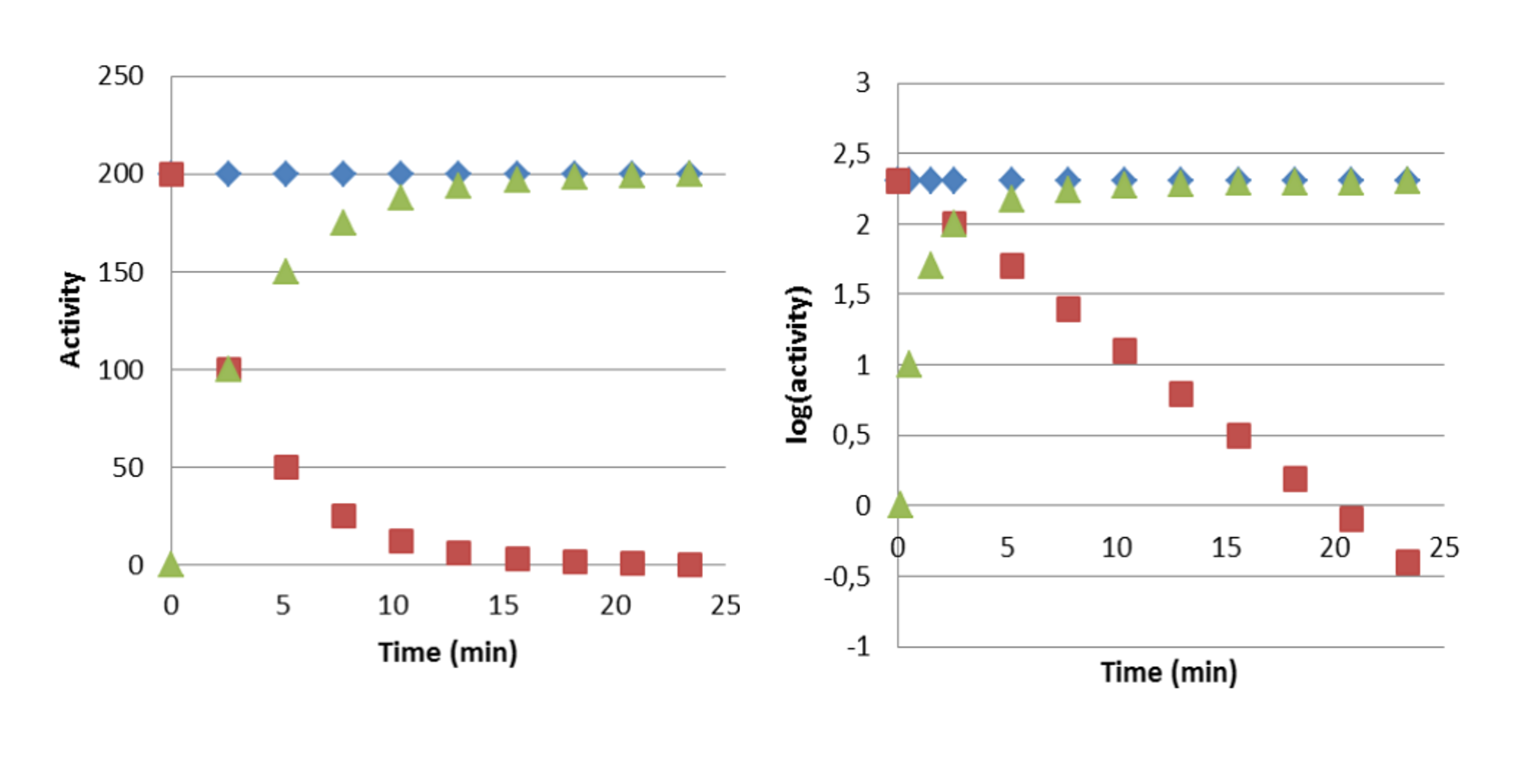 Figure VI.4. Development of a secular radioactive equilibrium in which the half-life of the parent nuclide very long and the half-life on the daughter nuclide (137mBa, t½ = 2.6 min) is considerably shorter than that of the parent (137Cs, t½ = 30 a). Left: activity on linear scale. Right: activity on logarithmic scale. Blue: 137Cs. Red: 137mBa if separated from 137Cs. Green: ingrowth of 137mBa with 137Cs after separation of 137mBa from 137Cs.
Figure VI.4. Development of a secular radioactive equilibrium in which the half-life of the parent nuclide very long and the half-life on the daughter nuclide (137mBa, t½ = 2.6 min) is considerably shorter than that of the parent (137Cs, t½ = 30 a). Left: activity on linear scale. Right: activity on logarithmic scale. Blue: 137Cs. Red: 137mBa if separated from 137Cs. Green: ingrowth of 137mBa with 137Cs after separation of 137mBa from 137Cs.
6.7.2. Transient equilibrium
An example of transient equilibrium is a beta decay chain where 140Ba decays to 140La and the latter to stable 140Ce.
$${}^{140}\text{Ba} \, (\beta^-, \, 12.8 \, \text{d}) \rightarrow {}^{140}\text{La} \, (\beta^-, \, 40.2 \, \text{h}) \rightarrow {}^{140}\text{Ce} \, (\text{stable})$$
[VI.XV]
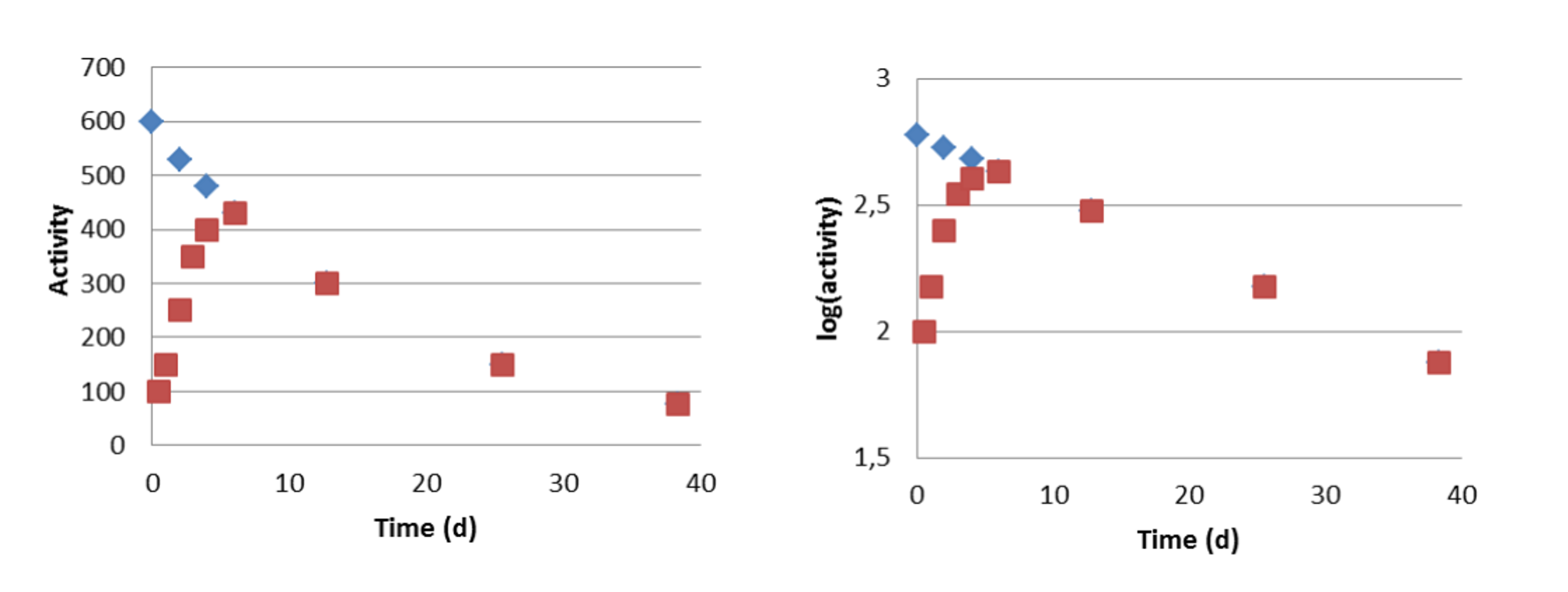 Figure VI.5. Development of a transient radioactive equilibrium in which the half-life of the parent (140La, t½ = 40.2 d) is so short that we observe decrease in its activity in a reasonable time and the half-life on the daughter nuclide (140Ba, t½ = 12.8 d) is shorter than that of the parent. Left: activity on linear scale. Right: activity on logarithmic scale. Blue: 140La. Red: 140Ba.
Figure VI.5. Development of a transient radioactive equilibrium in which the half-life of the parent (140La, t½ = 40.2 d) is so short that we observe decrease in its activity in a reasonable time and the half-life on the daughter nuclide (140Ba, t½ = 12.8 d) is shorter than that of the parent. Left: activity on linear scale. Right: activity on logarithmic scale. Blue: 140La. Red: 140Ba.
The transient equilibrium is otherwise identical with the secular equilibrium except that the parent nuclide decays with such a short rate that we observe decrease in activity in a reasonable time. After attaining the equilibrium in about ten half-lives of the daughter, about two weeks in case of Figure VI.5, both parent and the daughter decay at the rate of the parent nuclide. Also, after attaining the equilibrium the total activity is twice the activity of the parent nuclide.
6.7.3. No-equilibrium
An example of no-equilibrium case is the alpha decay pair 218Po (t½ = 3 min) → 214Pb (t½ = 26.8 min), where the half-life of the parent is shorter than that of the daughter. No equilibrium develops since the parent decays before the daughter.
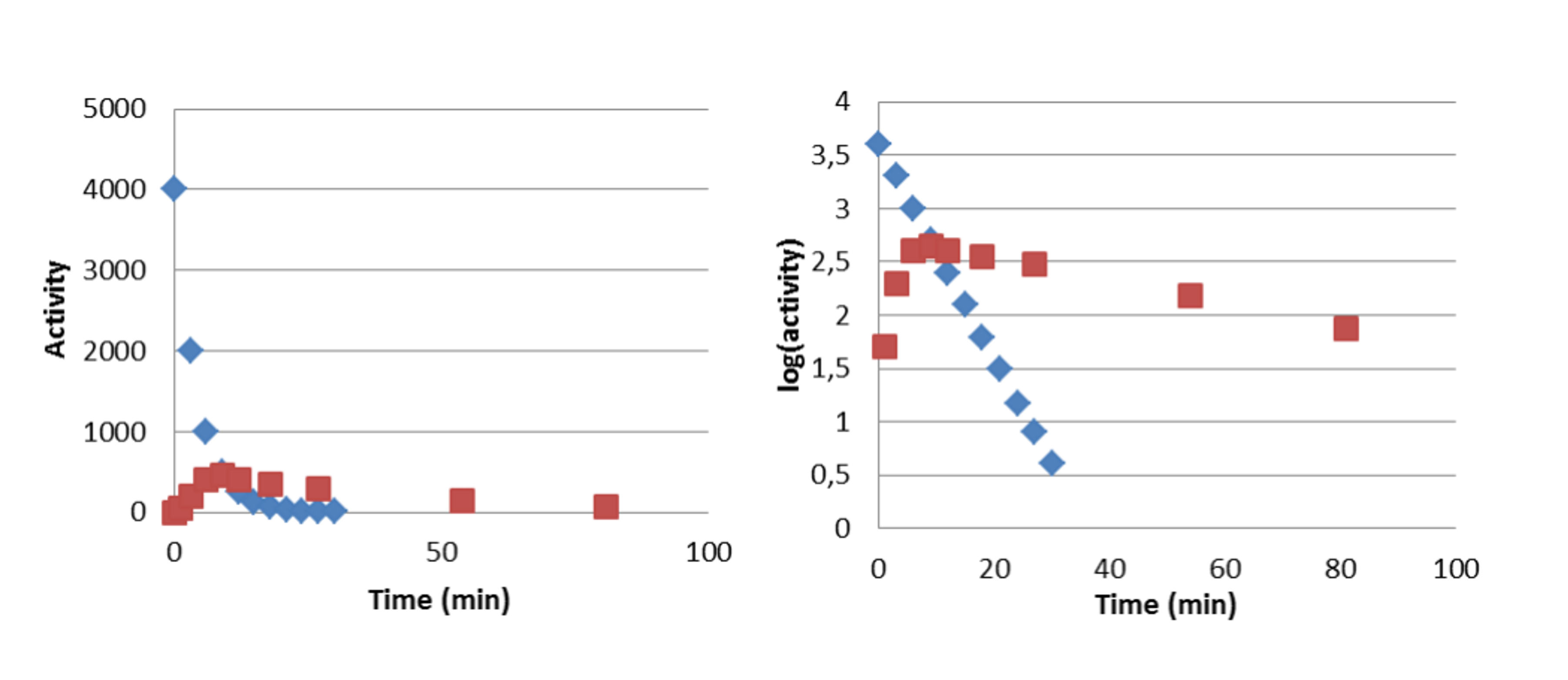 Figure VI.6. Development of activities in case of no radioactive equilibrium, in which the half-life of the parent nuclide (218Po, t½ = 3 min) is shorter than that of the daughter (214Pb, t½ = 26.8 min).
Left: activity on linear scale. Right: activity on logarithmic scale. Blue: 218Po . Red: 214Pb.
Figure VI.6. Development of activities in case of no radioactive equilibrium, in which the half-life of the parent nuclide (218Po, t½ = 3 min) is shorter than that of the daughter (214Pb, t½ = 26.8 min).
Left: activity on linear scale. Right: activity on logarithmic scale. Blue: 218Po . Red: 214Pb.
6.7.4. Equilibria in natural decay chains
In natural uranium and thorium decay chains there are individual pairs in which there would not be any equilibrium if they were present separately. An example of such pairs in the 238U decay chain is 234Pa parent (t½ = 6.7 h) and 234U daughter (t½ = 245000 y). They are, however, typically in equilibrium since the grandparent of 234Pa, 238U (t½ = 4.4 × 109 y), feeds continually new 234Pa and they are in equilibrium with each other. Since 238U has the longest half-life in the whole chain and therefore the activities of all subsequent radionuclides in the chain have the same activity as 238U supposing that the system has been closed millions of years. In the nature there are chemical processes, such as dissolution into groundwater, that remove some component of the chains which causes disequilibria in the chains.
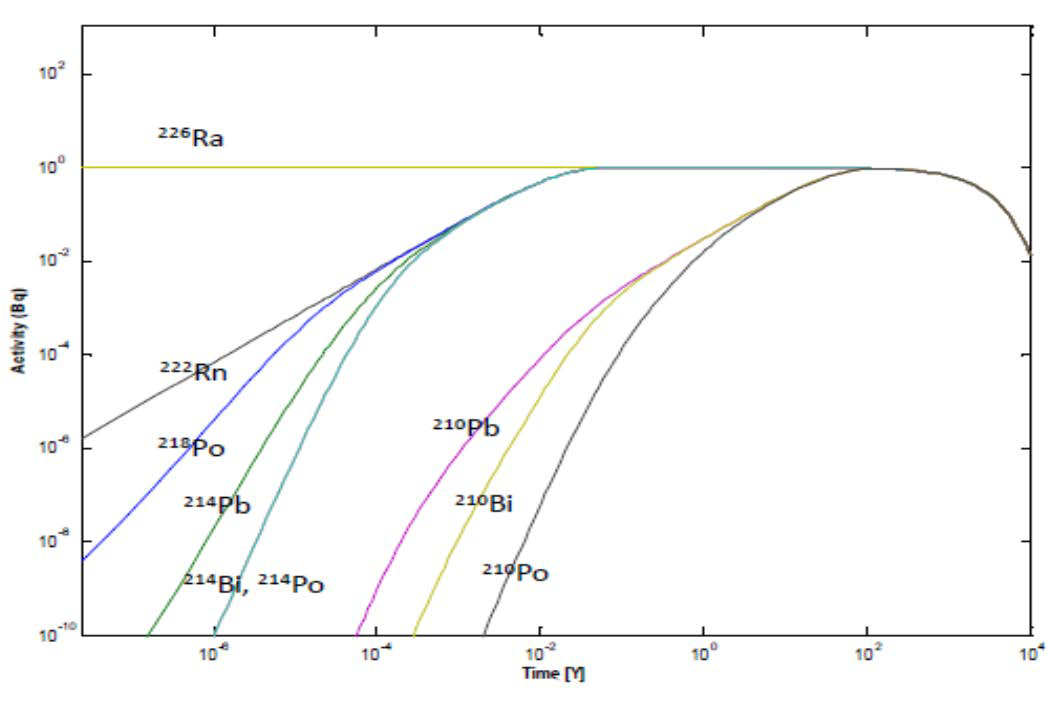
In the geosphere in the natural decay chains beginning from 238U, 235U and 232Th the activities of all members are the same in each series, identical with those of 238U, 235U and 232Th, in systems which have been preserved without disturbances long enough. In such case the series is in equilibrium state. If some component of the series is removed, by dissolution for example, the equilibrium is disturbed and a disequilibrium state is created. If for example uranium is dissolved from a primary uranium-bearing mineral by oxidation the remaining radionuclides in the series will be supported by its most long-lived radionuclide which is 230Th in case of 238U series. If the dissolved uranium will then be precipitated somewhere out of the system a new equilibrium will start to develop. The time required to attain the equilibrium is governed by the most long-lived daughter radionuclide in the series, 230Th in case of 238U series. The half-life of 230Th is 75000 years and this time is required to attain 50% of the equilibrium, 150000 years for 75% equilibrium, 225000 years for 87.5% equilibrium and eight half-lives, 600000 years, for 99.6% equilibrium. The disequilibria can be utilized in dating geological events. If for example, the 230Th/238U ratio is 0.5 in a uranium mineral we may calculate that this uranium mineral was precipitated 75000 years ago.
To calculate activities of all members in a series manually is a cumbersome task. Computer programs for this purpose have been fortunately developed. One of them is the Decservis-2 program developed at the Laboratory of Radiochemistry, University of Helsinki, Finland. An example of such calculation carried out by Decservis-2 is shown in Figure VI.6. Here, we assume separation of 226Ra (1 Bq) from the system and development of equilibrium between 226Ra and its progeny in 10000 years. We have to assume that the gaseous 222Rn, the daughter of 226Ra, is not escaped from the system. In the first phase, up to about a month, the equilibrium is attained with 222Rn, 218Po, 214Pb, 214Bi and 214Po and the time required for equilibrium is governed by the most long-lived member of these, 222Rn with a half-life of 3.8 days. In the second phase, up to about 200 years, the equilibrium is attained with 210Pb, 210Bi and 210Po and the time required for equilibrium is governed by the most long-lived member of these, 210Pb with a half-life of 22 years. The half-life of 226Ra is 1600 years and decrease in its activity and correspondingly activities of its progeny can be seen after about 1000 years.
Figure VI.7. Attainment of radioactive equilibrium of 226Ra progeny.
textbook/nrctextbook/chapter6.txt · Last modified: 2025-05-07 12:01 by Merja Herzig
A-CINCH Consortium
email: mst@evalion.cz | tel: +420 224 358 331 | Copyright © 2021 A-CINCH
This project has received funding from the Euratom research and training programme 2019–2020 under grant agreement No. 945301.