Sidebar
textbook:nrctextbook:chapter7
This is an old revision of the document!
Table of Contents
7. Interaction of radiation with matter
Chapter 7 from BASICS OF NUCLEAR PHYSICS AND OF RADIATION DETECTION AND MEASUREMENT - An open-access textbook for nuclear and radiochemistry students by Jukka Lehto
This chapter will address the immediate physical effects in medium arising from radiation resulting from radioactive decay and from other ionizing radiation. Later, in chapter XV we discuss on nuclear reactions, which also are a form of radiation interaction process in medium. The main focus in nuclear reactions will be production of radionuclides by intensive, high-energy particle beams from various accelerators and neutron-induced reactions in reactors. The chemical effects, such as the breaking of chemical bonds caused by radiation and formation of new ones, which fall within the scope of the radiation chemistry, are not dealt with.
Physical radiation interactions in the medium are important for many reasons:
- radiation cannot be detected and measured directly, but through the interactions of radiation with detector materials
- they are the primary cause of harmful effects of radiation on humans
- they are the basis for radiation protection measures
- they are the foundation of the radiation exploitation, such as production of radionuclides or autoradiography
The primary interaction mechanisms of radiation with medium are:
- scattering from the nucleus or electron shell
- excitation of nuclei or atoms
- formation of electromagnetic radiation (bremsstrahlung, Cherenkov radiation)
- absorption into the nucleus – nuclear reaction
Radiation other than the neutron radiation has a much greater possibility of interacting with the electron cloud than with the nucleus due to the much larger size of the electron cloud compared to nucleus. The removal of electrons from the electron shells of the medium atoms by ionization is the central pattern by which all radiation except neutrons loses their energy when moving in the medium. While the cross section of the ionization by protons or alpha particles can be several hundreds of thousands of barns (for definition, see Chapter XV) it is only under ten for nuclear scattering and still considerably less for nuclear transformations. Radiation, which causes ionization, is called ionizing radiation. The primary result in ionization is the formation of ion pair, electron and positive ion. In most cases, the emitting electrons are so high in energy that they can cause further ionization, secondary ionization, which can be an even a larger portion of the overall ionization than the primary ionization. The radiation energies generated by radioactive decay are typically at least in the keV range. These are high energies compared to energies of atom ionization, which are usually less than 15 eV and those of chemical bonding, which are even lower at 1-5 eV. It is therefore understandable that electrons arising from primary ionization have such a high kinetic energy to cause secondary ionization. Similarly, it is understandable that the primary high energy of a particle or gamma ray does not lose its energy in only one collision with an electron, but several.
7.1. Absorption curve and range
The ranges, length of passage, of different types of radiation in different type of media have been studied by determining absorption curves. Different thickness absorption plates are placed between a radioactive point source and a detector and the decrease in count rate in the detector is recorded as a function absorber thickness (Figure VII.1). All other factors than the absorber thickness, affecting the counting efficiency should be equal during the measurements. A graph of the count rate is then drawn as a function of absorber thickness, yielding an absorption curve of the radiation in the used absorber medium (Figure VII.2).
Figure VII.1 Radiation absorption curve determination system (modified from
https://tap.iop.org/atoms/radioactivity/511/page_47096.html).
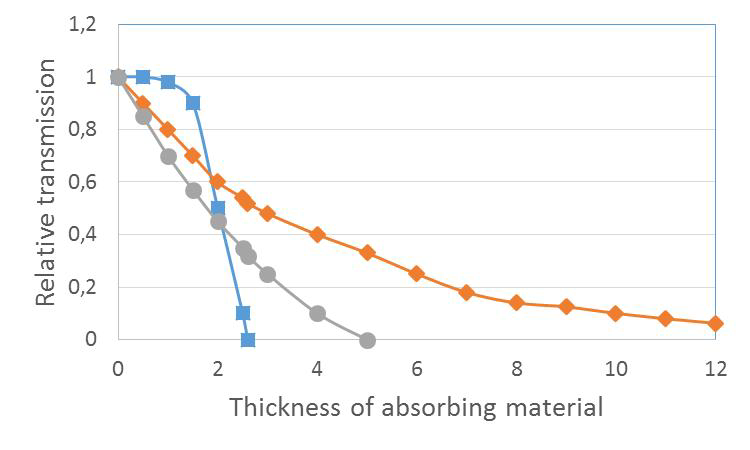
Figure VII.2. Absorption curves of alpha (blue), beta (grey) and gamma/neutron radiation (orange).
The absorption curves can be used to calculate the specific ranges of different radiation types. Range, for example, can be reported as the medium range, i.e. absorber thickness, in which the radiation intensity (counting rate) has dropped to half its original. Another way is to represent the maximum range, i.e. absorber thickness, wherein the radiation intensity has dropped to zero. Maximum range is a reasonable term for charged particle radiation, since indeed their intensity drops to zero. For gamma radiation and neutrons, as seen from Figure VII.2, however, it is not a suitable term since the decrease of gamma and neutron intensity decreases in an exponential manner and, in principle, no zero is reached. The range can be expressed in terms of absorption plate thickness, but it is more commonly represented as surface density (F), which is absorption plate thickness (d) multiplied by absorption material density (σ), in other words $F = d \times \sigma$. Surface density is used instead of absorber thickness to obtain a quantity which is independent of the nature of the absorber material. When using different types of absorber materials varying amount of electrons are present in the same thickness, less in case of low density material and vice versa. This is essential since the electrons are mainly responsible for radiation absorption. The number of electrons per unit mass (g) is, however, approximately the same for all elements at about 2.9×1023. Thus, when we express the absorber thickness as the surface density the radiation meets the same amount of electrons in its path in media at same surface density values, no matter what is the nature of the material. The most commonly used surface density unit is mg/cm2.
7.2. Absorption of alpha radiation
In comparison to other radiation types from radioactive decay, alpha radiation is characterized by the fact that the alpha particles are large and their energies are always high, usually between 4-9 MeV. Due to this, alpha particles do not readily scatter from medium atoms, rather their range is short and path is direct (Figure VII.3). For example, the 4.8 MeV alpha particles of 226Ra have a maximum range of 3.3 cm in air and only 0.0033 cm in water. Alpha radiation causes very intense ionization, for example, when traveling in air a 7.7 MeV alpha particle causes 3200 ion pairs/cm. The ion pairs generated in unit length is called specific ionization. Figure VII.4 shows specific ionization of alpha radiation (and of protons and electrons) as a function of particle energy. First specific ionization somewhat increases, but at energies higher than 1 MeV specific ionization decreases systematically. The specific ionization of alpha particles is clearly higher than that of protons, let alone electrons. This is due to their larger size and higher electric charge. Most of the electrons produced in primary ionization have a high energy, on average 100 eV, but some even higher than 3 keV and thus they cause strong secondary ionization.
Figure VII.3. Alpha radiation tracks of a 226Ra source imaged in a cloud chamber. (https://simple.wikipedia.org/wiki/Cloud_chamber).

Figure VII.4. The specific ionization of alpha particles, protons, and electrons (ion pair/mm) in the air as a function of particle energy.
Specific ionization is not uniform along the entire path traveled by the alpha particles. Ionization increases as the particle slows and reaches its maximum before it completely loses its energy and its positive charge (Figure VII.5).
Figure VII.5. Specific ionization of alpha particles and protons as a function of their residual range.
7.3. Absorption of beta radiation
In principle, beta radiation loses energy by the same processes as alpha radiation: ionization and excitation of medium atoms, but the essential difference is that the beta radiation range is much larger than that of alpha radiation and the track is not straightforward but quite winding. This is due to the fact that the beta particle size is much smaller than the alpha particle and hence, the probability of collision per unit length is lower. The small mass of the beta particle also means that the velocities are much greater than those of alpha particles. When the energies of the particles are the same, their velocities are proportional to their masses in accordance to the formula $E = m \times v^2$. As noted earlier, the maximum range of the 4.8 MeV alpha radiation is 3.3 cm in the air and only 0.0033 cm in water. Electrons with equivalent energy have maximum ranges that are far greater, e.g. 17 meters in the air. For alpha radiation, each particle travels approximately the same distance. In beta radiation, however, the track varies very much from one particle to another. The attenuation of a beta particle flux means that as the beta radiation flux travels further in an absorbing medium it loses a growing number of its individual particles.
Since the size and mass of beta particles and electrons in atoms are identical, the beta particles may lose a large fraction of their kinetic energy in individual collisions. In addition, their travel direction may change a lot, scattering can even occur in the completely opposite direction. The relative energy loss and change of path depend on both beta particle energy and the collision angle. The two identical particles, the beta particle and the shell electron, behave like billiard balls when one hits the other. The smaller the collision angle the greater is the change in residual path. As the beta particle hits the electron directly to its middle point, the ionized electron travels to the initial direction of the beta particle while the beta particle goes to opposite direction. The relative energy loss is higher for low energy beta particles. High energy beta particle paths are straightforward, for example even at energies of 0.2 MeV the path is straight. Beta particles with high energy will eventually slow down and their paths will become winding. The path of a beta particle beam is also affected by secondary electrons created by ionization, which cannot be distinguished physically from the original β--particles, emitting in varying directions and causing further ionization. In fact,70-80% of total ionization is caused by secondary ionization. Since the range of beta radiation is longer than alpha radiation, the specific ionization it causes is of a significantly lower magnitude (Figure VII.4).
Due to the above factors, as well as the fact that the beta particle energies are not constant,but vary between 0 and Emax, their absorption curve resemble an exponential curve (Figure VII.2). The absorption curve of a monoenergetic electron beam has a different shape, but if their energy is the same as the maximum energy of beta radiation, both cases yield approximately the same maximum range.
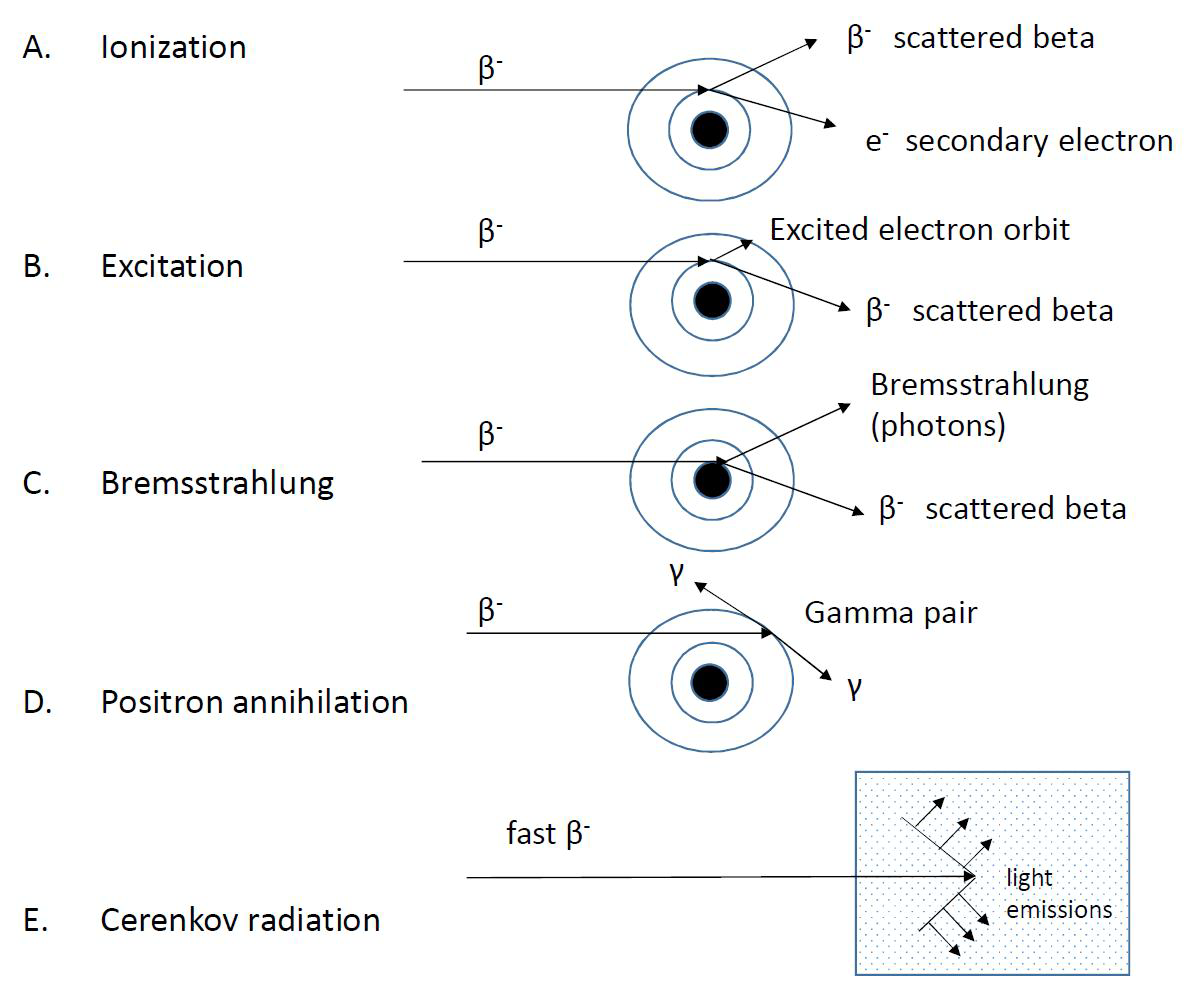
Figure VII.5. shows the five processes involved in beta radiation absorption:
Figure VII.5. Beta radiation absorption processes
As already stated, the beta radiation created in radioactive decay loses its energy in media by essentially two mechanisms: ionizing and excitation. Both processes cause approximately the same fraction of energy loss. In ionization a beta particle collides with a media electron, removes it from its orbit and proceeds with lower energy and to a direction different from that before the collision. In excitation, collision energy of beta particle is not enough for electron removal from an atom, but rather moves the electron to a higher energy level, i.e. yields electron excitation. The result of both processes is the emission of electromagnetic radiation, when an electron hole is filled by an electron from an upper electron shell or when an excitation level relaxes.
Bremsstrahlung is the electromagnetic energy that is generated when an electron interacts with the electric field of an atomic nucleus. The beta particle energy decreases by the amount of energy of the generated photon. The proportion of energy loss of beta radiation caused by bremsstrahlung is, however, very small. For example, only 1% of the energy of the 1 MeV beta particles is absorbed in aluminum by bremsstrahlung and the remaining almost exclusively by ionization and excitation. At higher beta energies the proportion of energy loss by bremsstrahlung increases. In addition, formation of bremsstrahlung is affected by the atomic number of the radiation absorbing material: the higher it is the more bremsstrahlung. For example, in lead already 10% of the energy of 1 MeV energy beta radiation is absorbed by formation of bremsstrahlung. Since the electromagnetic radiation of bremsstrahlung is noticeably more penetrating than beta radiation it is sensible to use a lower atomic number than lead as a protective material. One centimeter thick Plexiglas, for example, prevents penetration of high energy beta particles without the fundamental formation of bremsstrahlung like with lead.
When determining the absorption curve for beta radiation, a curve in accordance to Figure VII.6 is obtained by drawing an absorption layer thickness as a function of gross count rate measured from a beta source. After a specific absorber thickness is achieved the count rate levels off. This flat proportion is due to both the background radiation and the bremsstrahlung generated in the absorber. When their contribution is deducted from the total curve the beta radiation decrease due to the absorber and its maximum range are obtained (In Fig. VII.6 at 300 mg/cm2).
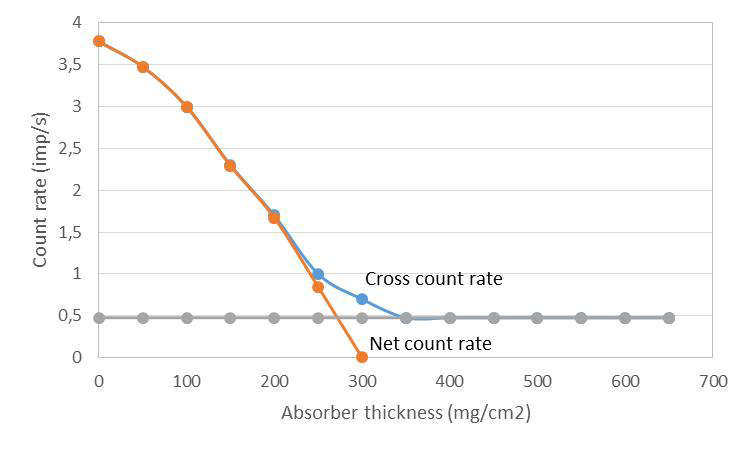 VII.6. Beta radiation absorption curve, background radiation and bremsstrahlung subtraction, as well as maximum range determination.
VII.6. Beta radiation absorption curve, background radiation and bremsstrahlung subtraction, as well as maximum range determination.
Positron particles experience the same interactions in the media as β- particles. When a positron has lost its kinetic energy, it combines with its antiparticle electron and they both disappear, annihilate.
$$e^- + \beta^+ \rightarrow 2\gamma$$
[VII.I]
The result is the development of two gamma photons emitting in opposite directions, at the same energy of 0.511 MeV. This energy is equivalent to the electron rest mass of 0.000548597 amu: $0.000548597 \, \text{amu} \times 931.5 \, \text{MeV/amu} = 0.511 \, \text{MeV}$. Upon the filling of the electron shells also X-ray radiation is generated.
Cherenkov radiation is blue light, which is created when a beta particle travels through the medium faster than light. In water the beta particle energy must be at least 263 keV to exceed the speed of light. In the absorption of beta radiation energy the formation of Cherenkov radiation forms only a small fraction, less than 0.1%. Cherenkov radiation may, however, be used to measure high energy (Emax >700 keV) beta radiation with liquid scintillation counter: this involves direct measurement of the light intensity of Cherenkov radiation without using liquid scintillator agents.
7.4. Absorption of gamma radiation
Since gamma radiation is weightless and uncharged, it rarely interacts in media. That is why it is penetratable and has a long range. Specific ionization of gamma radiation is small compared to beta radiation, let alone alpha radiation. For example, a 1 MeV gamma photon causes only one ion pair per centimeter in the air, compared to many tens by beta radiation and several tens of thousands by alpha radiation.
Gamma rays do not have an exact range. An individual gamma photon can lose its energy partly or completely in one or a few collisions with target atoms. When looking at a large number of gamma photons, or a flux, the attenuation, that is the flux density decrease, occurs exponentially according to the following formula:
$$\phi = \phi_0 \times e^{-\mu \cdot x}$$
[VII.II]
in which 𝜙0 is the initial flux density, 𝜙 the flux density, when gamma radiation has traveled through the absorption layer with a thickness of 𝑥, and 𝜇 is the absorption material’s total attenuation coefficient. Since attenuation is exponential, exact numerical range value cannot be obtained with gamma radiation. Instead, for example, each of the absorber material layer thicknesses, in which the flux density is reduced by e.g. half or one-tenth of the original, can be used as range values:
$$x_{1/2} = \frac{\ln 2}{\mu} \quad \text{and} \quad x_{1/10} = \frac{\ln 10}{\mu}$$
[VII.III]
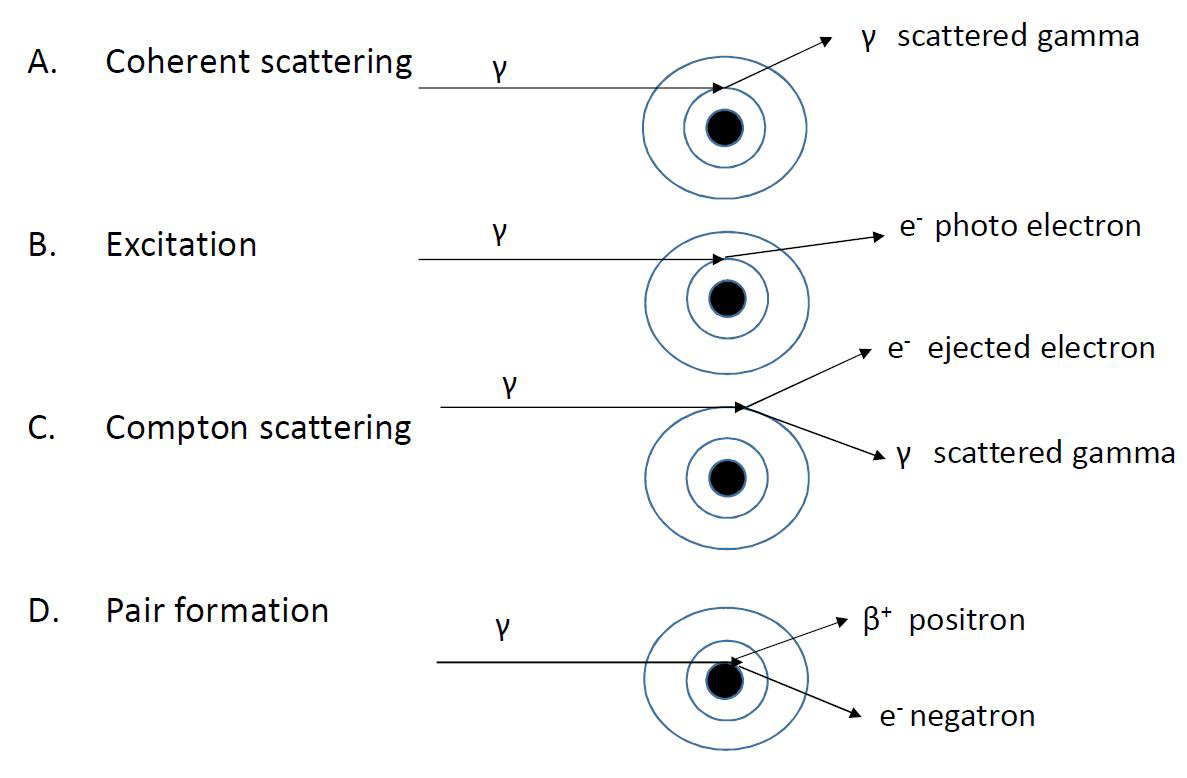
The total attenuation coefficient contains all of the interaction processes affecting gamma radiation attenuation. Overall, there are five interaction processes:
Figure VII.7 shows the first four of these interaction processes. The fifth, photonuclear reaction, which has a very small role in the overall attenuation, is covered in the chapter dealing with nuclear reactions. Coherent scattering, where the media atom absorbs the gamma photon and emits it again, also has little effect in energy loss of gamma radiation. The direction of the photon changes during scattering, but the energy is only reduced by the portion belonging to the recoil energy of the scattering atom. As the mass of the atom is large compared to the relativistic mass of the gamma photon (E = m×v2), its recoil energy is very small.
Figure VII.7. Attenuation mechanisms of gamma radiation in media.
The three other interaction processes, photoelectric effect, Compton scattering, and the pair formation, however, all have a large impact on the gamma radiation absorption.
In the photoelectric effect the gamma photon interacts with an individual orbital electron to which all of its energy is transferred and it is emitted from the atom. The kinetic energy of the emitted electron equals the kinetic energy of a gamma photon minus the electron binding energy. Generally, in the photoelectric effect the electron released is from the inner orbit. The filling of the vacant electron hole by higher energy orbit electrons causes formation of X-ray radiation and Auger electrons.
In Compton scattering, only part of the gamma photon energy transfers to the emitting electron. Gamma photon energy decreases by the electron binding energy and kinetic energy of the emitted electron. The scattered gamma photon continues traveling with less energy and change of direction. This scattered photon can still cause new Compton electron emissions.
In pair formation, the gamma photon is transformed by the action of nuclear electric field to an electron-positron pair. The phenomenon is the opposite of positron annihilation. Since the rest masses of electron and positron both correspond to energy of 0.511 MeV, the energy of the gamma photon has to be at least 1.022 MeV in order to form a pair. The rest of the photon energy will be shared equally as kinetic energy of the electron and positron:
$$E_{\gamma} = 1.02 \, \text{MeV} + E_{e^-} + E_{e^+}$$
[VII.IV]
The generated electron is absorbed by the media, as described in beta radiation absorption, and upon loss of its kinetic energy the positron is annihilated.
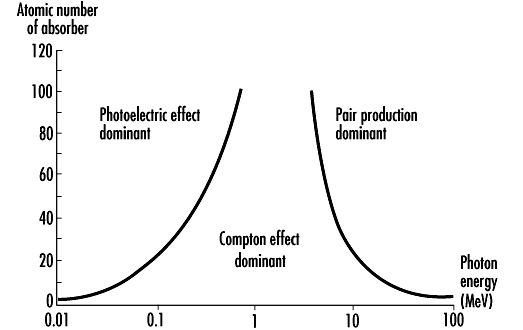
The fractions of these three interaction mechanisms in gamma radiation attenuation depend on two factors: gamma photon energy and media density (atomic number) (Figure VII.8). The photoelectric effect is prevalent at the lower gamma energies, the Compton scattering at the intermediate energy levels, and pair formation at high gamma energy levels. The increase of the atomic number of the absorber increases the fraction of photoelectric effect, as well as the probability of pair formation. The effect of the atomic number on Compton scattering is opposite, namely the probability decreases as the atomic number increases.
Figure VII.8. The effect of the gamma photon energy and atomic number of the absorber material on the gamma absorption by photoelectric effect, Compton scattering, and pair formation (http://www.ilocis.org/documents/chpt48e.htm).
7.5. Summary
| Type of radiation | Specific ionization in air (ion pair/cm) | Range in the air | Interaction process |
|---|---|---|---|
| Alfa radiation | tens of thousands | a few centimeters | • ionization • excitation |
| Beta radiation | tens to hundreds | a few meters | • ionization • excitation • bremsstrahlung formation • positron annihilation • Cherenkov radiation |
| Gamma radiation | few | exponential attenuation, “range” meters, tens of meters | • coherent scattering • photoelectric effect • Compton scattering • pair formation • photonuclear reaction |
textbook/nrctextbook/chapter7.1743511670.txt.gz · Last modified: 2025-04-01 14:47 by Merja Herzig
A-CINCH Consortium
email: mst@evalion.cz | tel: +420 224 358 331 | Copyright © 2021 A-CINCH
This project has received funding from the Euratom research and training programme 2019–2020 under grant agreement No. 945301.