Table of Contents
8. Measurement of radionuclides
Chapter 8 from BASICS OF NUCLEAR PHYSICS AND OF RADIATION DETECTION AND MEASUREMENT - An open-access textbook for nuclear and radiochemistry students by Jukka Lehto
There are two principal means to measure radionuclide activities: radiometric and mass spectrometric. Radiometric methods are based on detection and measurement of radiation emitted by radionuclides whereas in mass spectrometric methods number of atoms are counted. The results of these methods, activity (A) in case of radiometry and number of radioactive atoms (N) in mass spectrometry, can be can be converted to each other by the radioactive decay law equation
$A = N \times \frac{\ln 2}{t_{1/2}}$ where t½ is the half-life of the radionuclide. This chapter mostly discusses the basic principles of radiometric methods and at the end mass spectrometric methods are shortly described.
Detection and measurement of radiation are based on atomic scale interactions of particles and rays, emitted in radioactive decay, with detector materials. There are many types of interaction processes, as was discussed in chapter 7, but radiation detection and measurements make use of two of these processes, ionization and excitation. The electrons obtained in ionization are amplified to observe pulses representing individual decay processes. In the case of excitation light is formed in de-excitation process. These light photons are transformed into electrons which are further amplified to detectable electric pulses. In both cases the pulse rate is proportional to the decay rate and typically the pulse height to the energy of the detected particle or ray. Thus, each pulse obtained from the radiation measurement system represents individual radioactive decay event and the pulse height the energy of detected particle or ray.
8.1. Count rate and factors affecting on it
Radiation measurement system counts electric pulses resulting from primary interactions of radiation with the detector material, ionization and excitation. The primary result observed in radiation measurements is the number of pulses (X) which divided by the measurement time (t) gives the count rate (R) which in turn is proportional to the activity (A) of the source measured:
$$\frac{X(\text{imp})}{t(\text{s})} = R(\text{imp/s}) = E \times A(\text{Bq})$$
[VIII.I]
where E is the counting efficiency, i.e. the factor giving the fraction of particles or rays emitted in decay that were transformed into electric pulses in the measurement system. This counting efficiency depends on a number factors, coefficients (c) which except one decrease the count rate with respect to the [textbook:nrctextbook:chapter6#activity|activity]]:
$$E = c_{se} \times c_{ge} \times c_{dt} \times c_{bc} \times c_{ab} \times c_{sa}$$
[VIII.II]
where
• cse is the sensitivity coefficient representing the fraction of particles or rays, hitting the detector, which the detector is able to transform into electric pulses. Sensitivity factor is not only dependent on the detector material but also on type of radiation. All detector materials are more sensitive to alpha and beta radiation than to gamma radiation since large part of gamma radiation penetrates the detector. For example, in gas ionization detectors practically all beta and alpha particles entering the detector are transformed into electric pulses while only a few percent of gamma radiation.
•cge is the geometry coefficient which is relevant to all types of radiation in the same manner. In radioactive decay particles and rays emit randomly to all directions but only those hitting the detector can be detected. Considering a radioactive point source which is at a distance of h from a round-shaped detector with a diameter of r only those particles or rays emitted in the space angle $G=2π(1-sinα)$ can be detected (Fig. VIII.1). In this case the geometry factor is:
$$c_{ge} = \frac{G}{4\pi} = \frac{1}{2} (1 - \sin \alpha) = \frac{1}{2} \left(1 - \frac{h}{\sqrt{h^2 + r^2}}\right)$$
[VIII.III]
 Figure VIII.1. Effect of counting geometry on radiation detection of a point source.
Figure VIII.1. Effect of counting geometry on radiation detection of a point source.
In practice the situation is more complicated since the sources are seldom point sources. As a rule the geometry factor is the higher the closer is the source to the detector. To improve geometry in gamma spectrometry well-type detectors, instead of planar, are used. In these the source is placed inside a hole in the detector and a larger fraction of gamma rays are thus detected. The best geometry in obtained in liquid scintillation counting where the radionuclide is uniformly distributed in liquid scintillation cocktail and in principal all beta and alpha particles can lead to formation of light pulses when exciting scintillator molecules are surrounding them in all directions.
•cdt is the dead-time coefficient. Dead-time is the time when the detector is unable to process a new pulse as the processing of the former pulse is still ongoing. Thus, the dead-time is the minimum time that the detector needs to separate two radiation events and thus to be recorded as two separate pulses. The dead-time is measured from the equation:
$$R_0=\frac{R}{1-R\tau}$$
[VIII.IV]
where R is the observed count rate, τ the dead-time and Ro the count rate corrected for dead-time. The dead-time is the higher and the smaller is cdt the higher is the activity of the source measured which is visualized in Figure VIII.2. At high count rates the observed count rate is radically affected by dead-time and therefore should be taken into account.
Figure VIII.2. Observed count rate (R) as a function of count rate taking into account 10 µs dead-time of the detector.
• cbc is the backscattering coefficient relevant for beta particles. When scattering from atoms of surrounding matter beta particles considerably change their path direction and can be scattered even to opposite direction of their initial path. Some of the beta particles, not initially emitted towards the detector, may scatter from the surrounding material, such as a lead shield, and go to detector. Thus, backscattering can increase the observed count rate and it is the only factor in Eq. VIII.II that has a value higher than one. Backscattering is dependent on the backscattering material: the higher its atomic number is the more efficiently it scatters. Backscattering is also dependent on the structure of the measurement system: the closer to detector and source there is scattering material the higher is backscattering. Backscattering is relevant when measuring beta radiation with gas ionization detectors and semiconductor detectors but not when measurement is done with liquid scintillation counting. Gamma radiation and particularly alpha radiation do not scatter that much that it would be important in their measurement.
• cab is the absorption coefficient which counts for absorption of radiation between the source and the detector. It is dependent on the type and thickness of the matter between the source and the detector as well as on the type of radiation. The thicker the absorbing material and the higher is its density the larger fraction of radiation is absorbed in this material. Absorption is most relevant for alpha radiation (absorption of alpha radiation) since its range is short and it effectively absorbs even in air. Therefore, alpha measurements are done in vacuum. Measuring gamma radiation is least troublesome with respect to absorption since its range is long and it is readily penetrating radiation. The problem with beta radiation is between those of alpha and gamma radiation. When a beta measurement is done with liquid scintillation counting, part of the beta particle energies can be absorbed before they result in excitation with a scintillator molecule. When, in turn, beta measurement is done with a gas ionization detector beta particles can absorb in air between the source and the detector and especially in the window on the detector. With the high energy beta emitters, such as 32P (Emax = 1.7 MeV), this is not a problem but with low energy beta emitters, such as tritium (0.018 MeV), the absorption is already so high that gas ionization detectors are ruled out.
• csa is the self-absorption coefficient which is due to absorption of radiation into the sample itself. This factor is most important for alpha radiation and least important for gamma radiation while the importance for beta radiation is in between the two former. Since the range of alpha radiation is very short the counting sources in alpha spectrometry using semiconductor detectors are prepared as "massless" which means that the mass of the counting source should be as small as possible. The greater the mass of the source is the broader the peaks become, deteriorating energy resolution, and the lower the intensity of detected pulses is. Smallest mass is obtained by electrodeposition of alpha emitters on metal discs. Preparing counting sources by microcoprecipitation and counting the alpha spectrum from the resulting very small amount of precipitate on a filter somewhat deteriorates energy resolution but most often yields into a sufficient result. When measuring alpha radiation with liquid scintillation counting self-absorption is of no importance since alpha emitters are dissolved in the scintillation cocktail and practically all alpha particles yield formation of an electric pulse, i.e. the counting efficiency is nearly 100%. Gamma radiation is highly penetrating and for gamma rays of at least a few hundred keV energy self-absorption is of minor importance. For gamma rays of lower energy self-absorption has to be carefully taken into account. When measuring aqueous solutions self-absorption can be easily accounted for by measuring aqueous standard samples in same geometry as the unknown samples. For solid samples the standardization with respect to self-absorption, efficiency calibration, is not that simple since there is no comprehensive set of standards having various radionuclides in solid matrices identical or close to the composition of the unknown sample. Self-absorption in solid samples is dependent on many factors, most important of which are the elemental composition, density and thickness of the sample and the energy of gamma rays. When measuring beta radiation with liquid scintillation counting self-absorption is not a problem since the sample is usually dissolved in liquid scintillation cocktail. Only when measuring solid samples by liquid scintillation, self-absorption needs to be taken into account. However, when beta radiation is measured with gas ionization detectors self-absorption needs a careful consideration, especially when beta emitters with lower beta energies are measured and, therefore, the counting sources are prepared in a similar manner as in alpha spectrometry with semiconductor detectors, by electrodeposition or microcoprecipitation.
As seen, there are a number of factors affecting the counting efficiency. All of them have effect on the observed count rate. This, however, does not mean that the values of the coefficients would need to be determined in each activity measurement. Usually when the activity of an unknown sample (Ax) is to be determined its count rate (Rx) is measured and compared to the count rate obtained of a standard (Rst) with a known activity (Ast) and the activity of the unknown sample can be calculated as follows:
$$A_x=A_{st}\left( \frac{R_x}{R_{st}} \right)$$
[VIII.V]
This method, however, applies only when both the unknown sample and the standard are measured in identical conditions which guarantees same counting efficiency for both measurements. When measuring aqueous samples by gamma spectrometry it is enough that both the standard and the unknown sample are measured in the same geometry, i.e. the sample sizes are identical and the distance from the detector is the same. For solid samples, as already mentioned, this is usually not enough, since self-absorption needs to be taken into account either by using a standard with the same, or nearly the same composition, or by computational methods requiring knowledge on the chemical composition of the unknown sample. In liquid scintillation counting direct comparison of the count rate of the unknown sample to that of the standard is not used but the counting efficiency is determined for each individual sample. This, however, also needs standardization which is described in the chapter on liquid scintillation counting.
8.2. Pulse counting vs. energy spectrometry
In measuring radiation there are two modes:
- Pulse counting which means that pulses from a radioactive source are counted irrespective of their energy. Either pulses generated by all particles or rays are counted or they are counted at a defined energy range. Thus, in the pulse counting mode no multichannel analyzer is used.
- Energy spectrometry in which the energies of each particle or ray are determined and sorted to corresponding channels of a multichannel analyzer. As a result the number of pulses (or count rate when divided by measurement time) in each channel is obtained, i.e. an energy spectrum is obtained. Figure VIII.3 shows a simple example, the energy spectrum of 137Cs measured with a solid scintillation detector. The peak at the right corner represents pulses from the 662 keV gamma transition in the decay of 137Cs.
Figure VIII.3. Gamma spectrum of 137Cs measured with a solid scintillation detector.
Pulse counting is used to measure gross alpha and gross beta activities in environmental samples. In these no information is obtained on the radionuclide composition, which however, may in typical situations be approximately known. Pulse counting is also used in laboratory experiments with added tracers, for example when studying sorption of a certain radionuclide on a mineral in controlled conditions. In these, typically known amount of a single radionuclide is added to the system and its activity is measured after the experiments, for example in the solution phase in the above-mentioned sorption experiments. Since there are no interfering radionuclides present no energy spectrometry is needed. The activity of the tracer is measured by determination of pulses in the energy area (channel range) representing energies of the tracer. This type of measurements are typically carried out with gamma-emitting tracers using solid scintillation detectors. For example, to measure the activity of 137Cs tracer only peaks representing its photopeak are measured. This is done by setting discriminators to the pulses: the lower discriminator reject pulses with lower pulse size than set while the upper one reject those of higher than set. Thus amount of pulses from the set energy range is obtained and this can be converted to count rate by dividing with counting time. A third example of pulse counting mode is the measurement of beta radiation with GM-tubes. All beta particles entering the interior of the tube cause a pulse of same height. Thus no spectrometric data can be obtained.
If the sample contains several radionuclides and their individual activities are to be measured energy spectrometry is needed. Gamma spectrometers with semiconductor detectors, having very good energy resolution, can differentiate tens of radionuclides from the same sample and their activities are measured from the areas of specific peaks belonging to each nuclide. Solid scintillation detectors can also be used for energy spectrometry but are seldom used for that due to their limited energy resolution. Gamma spectrometry is also used for radionuclide identification in which positions of peaks and their relative intensities are made use of. Alpha spectrometry withsemiconductor detectors (alpha) is used to measure both activities of alpha emitters and their isotopic ratios, the latter bringing often valuable information, for example, on the source or origin of the alpha emitter. Alpha measurements, however, require radiochemical separations and typically only one element is measured at a time. Beta spectrometry is most often carried out by using a liquid scintillation counter, but also with proportional counter. Due to continuous nature of beta spectra, however, only one beta emitter is measured at a time. Sometimes it is possible to measure two beta emitters from the same samples if the energy difference of the two beta emissions is high enough.
8.3. Basic components of radiation measurement equipment systems
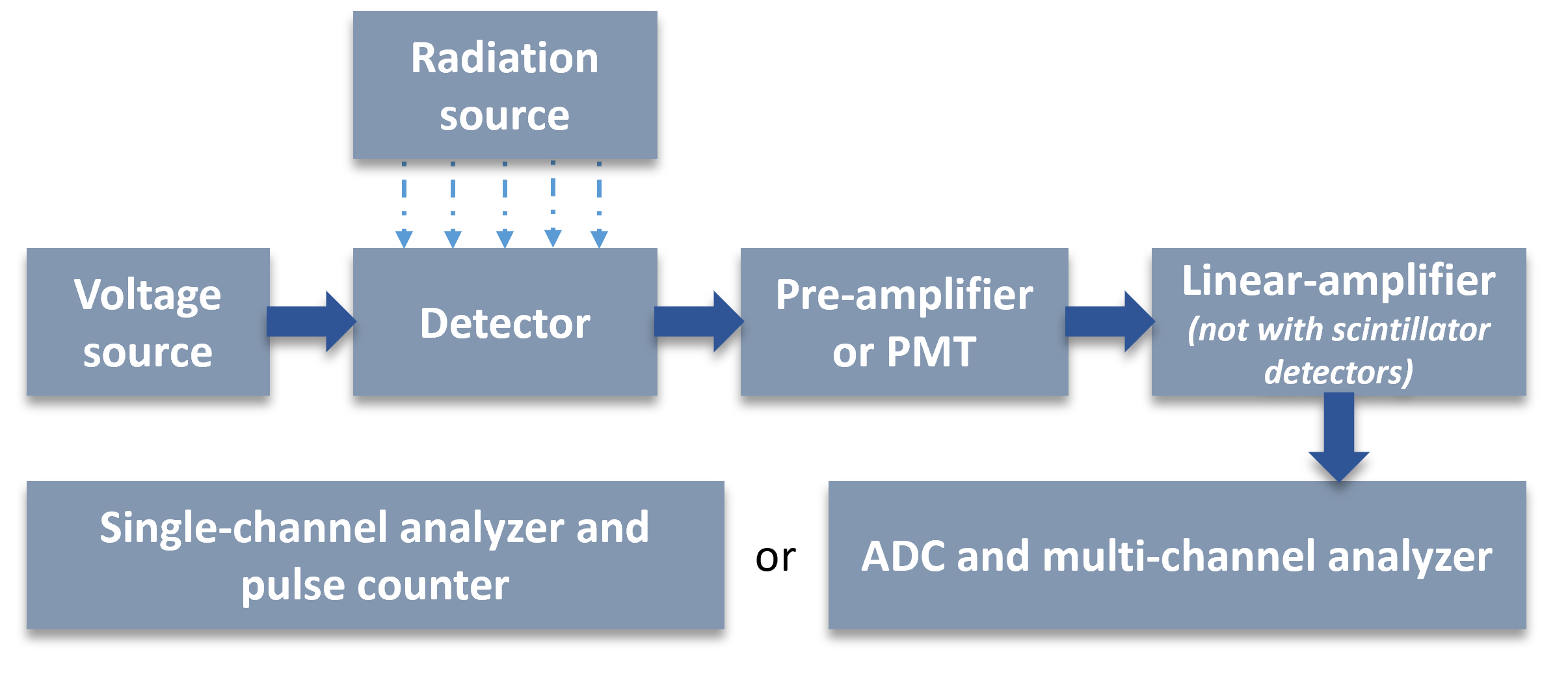
Radiation measurement equipment systems consist of the following components (Figure VIII.4):
- Detector, the function of which is to transform the energy of radiation into an electric pulse (gas ionization detectors and semiconductor detectors for gamma and alpha radiation) or to a light pulse (liquid scintillation detectors and solid scintillators ). Various detectors are discussed in later chapters in more detail.
- Voltage source which collects the initial electric pulses into electrodes.
- Preamplifier which amplifies the weak pulses coming from semiconductor detectors to enable the pulse transfer through cables into the main amplifier.
- Main amplifier is called linear amplifier since its function is not only to increase the pulse height to a measureable one but also preserve the energy information. This is done by amplifying each initial pulse with the same factor so that the observed pulse heights are linearly related to the heights of the initial pulses coming from the detector and the preamplifier.
- In scintillation detectors there is, instead of preamplifier and linear amplifier, a photomultiplier tube (PMT) which converts the light pulse into an electric pulse and amplifies the initial pulse into a measurable electric pulse.
After this there are two options depending on whether the equipment is used as a multichannel analyzer or as a single-channel analyzer. The former is used in energy spectrometry and the latter in pulse counting.
- A multichannel analyzer (MCA) sorts the pulses into various channels depending on their pulse height which is proportional to the energy of the particle or ray. For example 1 mV pulse goes to channel 1, 12 mV pulse to channel 12 and 715 mV pulse to channel 715. This results in the formation of an energy spectrum. A multichannel analyzer may have even thousands of channels. Prior to sorting the pulses into channels the analog-to-digital converter (ADC) transforms the analogical pulses into digital form.
- A single channel analyzer (SCA) counts only pulses at a defined height range. As described above, selection of pulse height range is accomplished with voltage discriminators, lower and upper. In addition, there is a pulse counter that sums all pulses coming to the discriminator window. For example, single channel can be set to count only pulses with heights between 50 mV and 150 mV, i.e. pulses that would go channels 50-150 in the multichannel analyzer, presuming same settings. Single-channel analyzer is used to measure only one radionuclide at the time. The discriminators are set by measuring the spectrum of the desired radionuclide by using a narrow discriminator window at increasing mV range. Plotting the counts at increasing mV results in the formation of an energy spectrum. The measurement window is set by measuring the spectrum of the desired radionuclide and selecting from the spectrum the lower and upper discriminator voltage values so that the pulses from the photopeak is between them. Single channel mode is typically used in gamma counters with solid scintillation detectors.
Figure VIII.4. Components and scheme of radiation measurement equipment systems. PMT is photomultiplier tube.
8.4. Energy resolution
In energy spectrometry it is essential that the measurement system can differentiate different particle or ray energies as efficiently as possible. This is mostly dependent on the type of detector. The better the energy resolution the better the system can differentiate energies close to each other and the narrower are the observed peaks in a spectrum. Resolution (R) is expressed as the peak width at half of the height of peak maximum (FWHM = full width at half maximum) (Figure VIII.5). Instead of the absolute value the energy resolution can also expressed as the relative value (ΔE/E)×100%, where E is the energy of the peak maximum and ΔE is FWHM. For example, for 137Cs the energy resolution of the 662 keV peak is typically 60 keV and the relative resolution value (60/662)×100% = 9%. For semiconductor gamma detectors, which are superior with respect to energy resolution compared to solid scintillation detectors the energy resolution is often expressed as the FWHM of the 60Co peak at 1332 keV. The energy resolution of semiconductor gamma detectors is clearly below 2 keV. Energy resolution is also dependent on the energy, the absolute values being better for low energy gamma rays, and therefore the resolution value should always refer to the energy for which is given. The energy resolution for 2 MeV gamma rays of germanium semiconductors is below 2 keV (0.1%), below 1.5 keV (0.15%) for 1 MeV rays and below 1 keV (0.2%) for 0.5 MeV rays. For NaI(Tl) solid scintillation detector the corresponding values are about 100 keV (5%) for 2 MeV rays, 70 keV (7%) for 1 MeV rays and about 50 keV (10%) for 0.5 MeV rays. Thus the germanium detectors have about 50-times better resolution compared to the NaI(Tl) detectors. Silicon semiconductor alpha detectors have resolutions between 20-30 keV (0.4-0.6% for typical alpha energies of 4-7 MeV) which are about ten times lower than values obtainable with liquid scintillation counters.
Figure VIII.5. Energy resolution of spectrum peak.
8.5. Radiation detectors and their suitability for the measurement of various types of radiation
Most typical radiation detectors discussed in following chapters can be divided into three groups:
1. Gas ionization detectors
2. Scintillation detectors
3. Semiconductor detectors (semiconductor detectors for alpha spectrometry and semiconductor detector for gamma spectrometry)
Alpha radiation is most accurately measured with semiconductor detectors. Their background pulses are very low and their energy resolution is good, at 5 MeV energies even 15 keV. The energy resolution of liquid scintillation counting, another choice to measure alpha radiation, is about ten times poorer than that of semiconductor detectors. Sample preparation for liquid scintillation counting is, however, clearly less difficult since the sample is just dissolved in the liquid scintillation cocktail for measurement while for semiconductor detectors counting sources need to be prepared by electrodeposition or microcoprecipitation. Another benefit of liquid scintillation counting is a very good, practically 100%, counting efficiency. In liquid scintillation counting the beta emitters present can cause problems by creating extra pulses to alpha peaks. In modern liquid scintillation counters this is overcome by alpha-beta discrimination system that differentiates alpha and beta pulses from each other and count them separately. Due to the poor energy resolution liquid scintillation counting is not a proper method to determine the isotopic ratios of alpha emitters. For this purpose semiconductor detectors need to be used.
For beta radiation the most often used method is liquid scintillation counting. It yields into high counting efficiencies and is suitable also for low energy beta radiation. Liquid scintillation counting also enables determination of beta spectra. Usually, however, due to the continuous nature of beta spectra, only one beta emitter can be measured at a time. Other options to measure beta radiation are the gas ionization detectors, GM tube and proportional counters, the latter of which can also produce beta spectra. The drawbacks of gas ionization detectors are more laborious counting source preparation, lower counting efficiency and the fact they cannot be easily used in measurement of beta emitters with the lowest energies, such as tritium. The benefit of gas ionization detectors is their clearly lower background compared to liquid scintillation counting and thus much lower detection limits are obtained.
For the measurement of gamma radiation solid scintillation and semiconductor detectors are used. The benefit of solid scintillation detectors is their higher counting efficiency as the detectors can be produced in large sizes and they are often of well-type in which the sample is inside the detector. Solid scintillation detectors are usually utilized for gamma counting in single-channel mode. The benefit of semiconductor detectors is their superior energy resolution compared to solid scintillators and therefore they are typically used for gamma spectrometric measurements in radionuclide identifications and measurement of radionuclide activities from samples having several gamma-emitting nuclides.
In a well-equipped radionuclide laboratory, measuring a range of radionuclides, there are the following radiation measurement apparatus:
- semiconductor detector(s) for gamma spectrometry for the determination of radionuclides from environmental and radioactive waste samples, for example
- gamma counter(s) having a solid scintillation detector and a sample changer for the measurement of tracer gamma emitters used in model experiments
- liquid scintillation counter(s) with alpha-beta discrimination for the measurement of tracer beta and alpha emitters as well as beta and alpha emitters separated from various samples
- low background liquid scintillation counter(s) for the measurement of low beta activities
- semiconductor alpha detector(s) for the measurement of alpha emitters separated from various samples
- gas ionization detector(s) to measure low activity beta emitters separated from various samples
8.6. Measurement of radionuclides with mass spectrometry
An alternative for radiometric methods for the determination of radionuclide activities is mass spectrometry. Most often inductively-coupled mass spectrometry (ICP-MS) is today used for this purpose. As mentioned, mass spectrometer counts atoms instead of radiation. This makes mass spectrometry particularly suitable for the measurement of long-lived radionuclides for which the detection limit of mass spectrometry is below those obtained by radiometric methods. For example, for 99Tc (t½ = 211000 y) the detection limit is at best 1 mBq for a gas ionization detector and clearly higher in liquid scintillation counting. 1 mBq means that there are about four decays in an hour but this activity of 99Tc corresponds to about 100 million atoms. This amount of atoms can be easily detected and counted by mass spectrometry and even as low as a 0.001 mBq detection limit can be achieved by ICP-MS. In principle all radionuclides with half-lives longer than 100 years can be measured by ICP-MS. However, for the radionuclides with half-lives round this limit, radiometric methods are still more sensitive and provide with more accurate results.
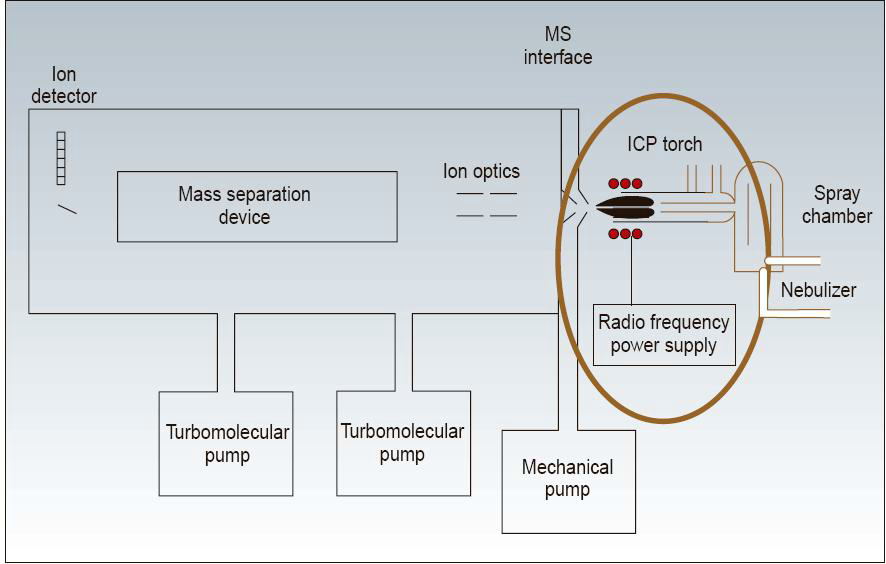
The components of an ICP-MS are presented in Figure VIII.6. The sample solution is introduced into the system by a nebulizer which turns the solution into a fine mist (aerosol). This is transferred with argon flow into the torch where plasma is created with the help of radiofrequency. Plasma atomizes the sample, ionizes the atoms and the ions are directed into a mass analyzer for the separation of ions based on their mass to charge ratio (m/z).
Figure VIII.6. Components of an ICP-MS system (http://www.people.fas.harvard.edu/~langmuir/SN-ICP-MS.html).
The mass analyzer is either quadrupole or double focusing system. The former is smaller, cheaper and easier to operate. The latter, however, is much more sensitive yielding to lower detection limits and to more accurate isotopic ratios. A quadrupole consists of four metallic rods aligned in a parallel diamond pattern. By placing a direct current field on one pair of opposite rods and a radio frequency field on the other pair, an ion of a selected mass and charge ratio (m/z) is allowed to pass through the rods to the detector while the others are forced out of this path. By varying the combinations of voltages and frequency, an array of different m/z ratio ions can be scanned in a very short time. The high-resolution double focusing system in turn consists of an electromagnet and an electrostatic analyzer in series. After mass separation the ions are detected and counted.
Some radionuclides, such as uranium, can be measured from natural waters directly with ICP-MS without chemical separation of interfering elements. Most radionuclides, however, need to be separated into a pure form prior to measurement. The separation requirements may essentially differ from those used in radiochemical separations for radiometric measurements. For example, if plutonium is measured by alpha spectrometry 1% of uranium activity in the counting source does not result in a large error. In mass, however, this 1% activity means about 2000-times excess of 238U compared to 239Pu which would prevent the measurement of plutonium. In mass spectrometry there are three types of interferences that need to be taken into account when measuring radionuclides. First, the isobars with approximately same mass cause interference, for example 129Xe in 129I measurement and 135Ba in 135Cs measurement. Second, in the plasma there are not only single atoms formed but also polyatomic ions such as 204Hg35Cl or 238UH which interfere with the measurement of 239Pu having approximately the same mass. Even though only a small fraction of the total elemental concentrations forms these polyatomic ions 204Hg35Cl or 238UH their concentrations are nevertheless much higher than that of plutonium due to the greater abundances of the polyatomic ion forming elements, in this case Hg, Cl and U. Therefore, chemical separations are needed to enable measurement of radionuclides at very low concentrations. Third type of interference comes from broadening the neighbor mass peaks at higher concentration. Due to this, for example, 238U at much higher concentrations compared to plutonium causes extra counts to the mass peak of 239Pu.
ICP-MS is increasingly used for the measurement of long-lived radionuclides, especially actinides. For neptunium it is clearly superior to radiometric methods due to its low specific activity. In plutonium measurement, mass spectrometry also offers a change to measure individually 239Pu and 240Pu which cannot be separated from each other in alpha spectrometry. In turn, 238Pu cannot be measured by mass spectrometry due to interference of uranium. Thus, to determine all relevant plutonium isotopes both mass and alpha spectrometry are needed.
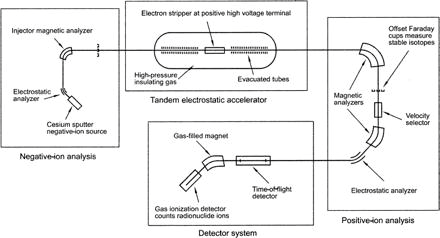
The most sensitive mass spectrometric method, even more sensitive than high-resolution ICP-MS, is accelerator mass spectrometry (AMS) which consists of two electromagnet mass analyzer and a tandem accelerator in between (Fig. VIII.7). AMS is very suitable for the measurement of long-lived radionuclides, such as 14C, 36Cl, 41Ca, 59Ni, 129I and actinide isotopes. For 14C measurement in carbon dating it has become a standard method. All radionuclide measurements with AMS require chemical separation of the target nuclide into a pure form.
Figure VIII.7. Components of accelerator mass spectrometry (AMS) (http://gsabulletin.gsapubs.org/content/115/6/643/F1.expansion.html).
The direct determination of radionuclides from solid samples can be accomplished in two ways: laser ablation ICP-MS and secondary ion mass spectrometry (SIMS). In laser ablation the solid sample is exposed to a laser beam and the elements thus evaporated from the surface are directed into ICP-MS for mass analysis. In SIMS the surface is sputtered with Cs+ ions and the elements released from the surface are directed into a mass analyzer.