Table of Contents
16. Production of radionuclides
Chapter 16 from BASICS OF NUCLEAR PHYSICS AND OF RADIATION DETECTION AND MEASUREMENT - An open-access textbook for nuclear and radiochemistry students by Jukka Lehto
This chapter deals with production of radionuclides for utilization in research, medical use and industrial use etc. Production of radionuclides, such as transactinides, for the study of their properties is not discussed here. Radionuclides are produced either in reactors or in particles accelerators, particularly cyclotrons. These two methods are complementary to each other both offering advantages over the other.
16.1. Production of radionuclides in cyclotrons
In cyclotrons nuclear reactions are induced by bombarding target atoms by proton-bearing particles, such as protons, deuterons and alpha particles. Depending on the bombarding particle and its energy various product nuclides are obtained (see Figure XVI.3). Most reactions result in change of the atomic number yielding product nuclei having a higher atomic number (or sometimes lower, for example in (d,α) reaction). These nuclei are proton-rich and therefore decay by positron emission or electron capture. Since the product nuclei are of a different element than the target nuclei cyclotrons can produce carrier-free radionuclides. This means that after chemical or physical separation the product nuclide does not contain any (or contains only very minor amounts) of stable isotopes of the same element. This results in the formation of product nuclides with very high specific activity (= activity divided by the mass of the product element). Use of carrier-free radionuclides is in many cases advantageous, for example in labelling of organic molecules, where as many positions in the molecules are aimed to label with a radionuclide and not with a stable isotope of the same element. In carrier-bearing radionuclide products the stable isotopes always “dilute” radionuclides and yield in lower specific activities. An example of cyclotron-produced radionuclides is 22Na which can be produced from magnesium with the reaction ${}^{24}_{12}\mathrm{Mg}(d,\alpha){}^{22}_{11}\mathrm{Na}$ (t½ = 2.6 a). Since the target is made of magnesium it does not, after chemical separation of sodium from magnesium, dilute the product.
Even high activities represent very low chemical amounts, for example 1 MBq of 22Na corresponds to only 2×10-10 moles or 4.5 ng of sodium which means that the specific activity is very high at 5×1015 Bq/mol or 2×1014 Bq/g. In practice completely carrier-free radionuclides are not achieved due to contamination. Furthermore, small amounts of carrier are often added to the system to prevent losses of the radionuclides due to adsorption, for example.
16.2. Production of radionuclides in reactors
As cyclotrons produce proton-rich radionuclides reactors produce neutron-rich ones. The reactions needed for radionuclide reactions are typically neutron capture reactions by using thermal neutrons. An example of such reactions is ${}^{23}_{11}\mathrm{Na}(n,\gamma){}^{24}_{11}\mathrm{Na}$ to produce 24Na (t½ = 15 h). As is seen from the reaction formula both the target and the product are of same element, sodium in this case. Thus all radionuclides produced in reactors using ($n, \gamma$) neutron capture reaction results in the formation of products containing carrier and therefore the specific activities of such radionuclides are fairly low. 24Na produced by neutron capture reactions has a specific activity of 1011 Bq/g Na at maximum, whereas 24Na produced from 26Mg in cyclotron by the ($d, \alpha$) yields a high specific activity of 1013 Bq/g Na. The neutron-rich radionuclide produced in reactors decay by beta decay (β-) to elements having a higher atomic number. In the case the desired radionuclide is a radionuclide produced in the β- decay of the primary product produced in a neutron capture reactions. Carrier-free radionuclides can be obtained after chemical separation. Radionuclides can be produced by reactors also by utilizing fission reactions, particularly thermal neutron induced fission of 235U. In this case the number of radionuclides produced is high and the required separations for desired radionuclide/s may be laborious and time-consuming.
Chapter XV describing nuclear reactions give the equations (XV.XI-XV.XVI) and Figure XV.2 presenting the kinetics of nuclear reactions used in radionuclide productions. These equations are needed to calculate the required irradiation times to produce a radionuclide with known half-life using a nuclear reaction with known cross section at given irradiation flux and bombarding energy.
16.3. Radiochemical and radionuclidic purity
When a tracer experiment with a certain radionuclide is done it is most often desirable that there are not any other radionuclides present since measurement of single radionuclide is easier as no radiochemical separations nor spectrometric analysis are needed. When a tracer product contains only one specific radionuclide, it is called radionuclidic pure. Radionuclide purity as a measure means the activity fraction of a specific radionuclide of the total activity. To produce radionuclidic pure tracers by nuclear reactions is not an easy task. The conditions in production reactions, particularly projectile energy and bombardment time, should be kept so that only one product nuclide is observed. This is, however, not typically possible since the cross sections of various reactions overlap in excitation function. For example, if 209Po tracer is produced in cyclotron by bombarding 209Bi with deuterons by the reaction ${}^{209}\mathrm{Bi}(d, 2n){}^{209}\mathrm{Po}$ the optimum deuteron energy of about 15 MeV, resulting in the highest yield, may not be used due to coproduction of 208Po. Instead, somewhat lower deuteron energies should be applied to minimize the fraction of 208Po (Fig. XVII.1).
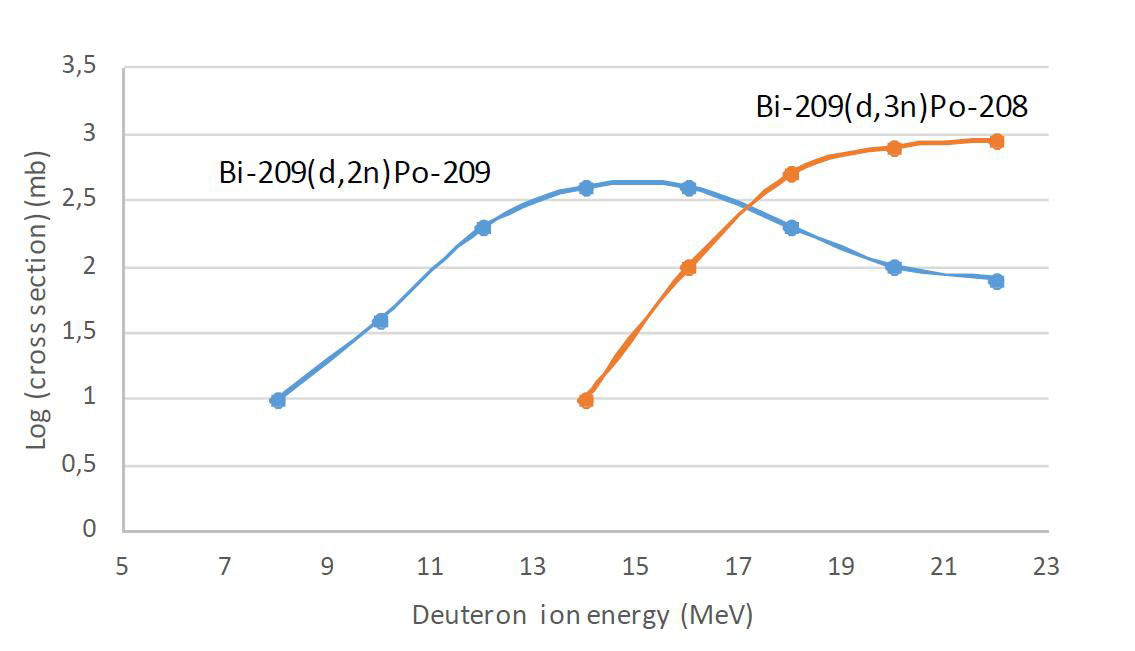 Figure XVI.1. Excitation function of 209Bi(α,xn)-reactions (data from Ramler et al. Physical Review 114(1959)154).
Figure XVI.1. Excitation function of 209Bi(α,xn)-reactions (data from Ramler et al. Physical Review 114(1959)154).
Another critical factor in producing radionuclidic pure tracers is the purity of the target material. Even very low amounts of impurities may result in considerable amounts of undesired radionuclides in the product, especially in case where the impurity atoms have higher cross sections for the used projectiles than the actual target atoms. To avoid formation of undesired radionuclides elementally very pure targets are typically needed. In some cases, elementally pure targets are not enough to prevent formation of undesired radionuclides, but even isotopically pure targets are needed. For example, in the production of 18F by the reaction ${}^{18}\mathrm{O}(p,n){}^{18}\mathrm{F}$ water enriched with respect to 18O is used as the target. The enrichment of 18O in the target water is about 97% while in the natural water it is only 0.2%. Isotopically pure target materials may be very expensive.
In addition to radionuclidic purity another term, radiochemical purity is important, particularly in labelling of organic molecules, for radiopharmaceutical purpose for example. Radiochemically pure compounds are the desired compounds containing the radionuclide or the compounds containing the radionuclide in a desired position. In, for example, 18F-labelled radiopharmaceutical 2-FDG (2-deoxy-2-[18F]fluoro-D-glucose) product the radiochemically impure compounds are those where the 18F-label is somewhere else than in 2-deoxy position or other 18F-labelled compounds, such as tetra-acetyl-2-[18F]FDG.
16.4. Radionuclide generators
Radionuclide tracers are commercially typically available as liquids containing radionuclides as ions, for example 137CsCl and NH4H32PO4 or as labelled compounds, such as [methyl- 14C]methionine and [35S]methionine. A number of radionuclide tracers are available in a mode of generator. In these a radionuclide, produced either in a reactor or a cyclotron, is trapped in column containing a sorbent, such as alumina, capable of sorbing this radionuclide. In the column the sorbed radionuclide decays to its daughter nuclide which is the desired tracer nuclide. The sorbent needs to efficiently trap the parent nuclide but not the daughter which should be eluteable out from the column while the parent nuclide remains. Another requirement is that the half-life of the daughter is shorter than that of the parent; otherwise no radiochemical equilibrium would be attained in the column.
As examples of radionuclide generators, the 99mTc and 137mBa generators are described. In a Tc generator the parent 99Mo (t½ = 66 h), produced in a reactor by neutron activation of stable 98Mo, is trapped in an aluminum oxide column where it decays to a short-lived 99mTc (t½ = 6.0 h). Tc forms an anionic TcO4- ion that can be eluted from the column with NaCl solution while 99Mo remains in the column as MoO42-. 99mTc is widely used as a radiopharmaceutical in hospitals in single photon tomography imaging of humans. 99mTc emits fairly energetic gamma rays (143 keV) which can be readily detected with gamma detectors.
In a 137mBa (t½ = 2.6 min) generator the parent 137Cs (t½ = 30 y) is trapped in transition metal hexacyanoferrate column, such as K2CoFe(CN)6. In the column the parent decays to 137mBa which can be eluted from the column with NaCl solution. Transition metal hexacyanoferrates are extremely selective for cesium and thus trap it very efficiently. 137mBa is used in monitoring of industrial processes for example in examining flow profiles on liquids in pipes. 137mBa emits energetic gamma rays (662 keV) which can be detected from outer surfaces of pipes, for example.
Table XVI.I Most important radionuclide generators.
| Mother nuclide | Decay properties | Daughter nuclide | Decay properties | Application |
|---|---|---|---|---|
| 41Ti | EC, γ; 47 y | 44Sc | β+, γ; 3.9 h | Teaching |
| 68Ge | EC; 288 d | 68Ga | β+, γ; 1.1 h | Medical |
| 87Y | EC, 3.3 d | 87mSr | γ; 2.8 h | Medical & teaching |
| 90Sr | β-; 28 y | 90Y | β-; 64 h | Heat/calibration source |
| 99Mo | β-; 66 h | 99mTc | γ; 6.0 h | Medical |
| 113Sn | EC, γ; 115 d | 113mIn | γ; 1.7 h | Medical |
| 132Te | β-, γ; 76 h | 132I | β-, γ; 2.3 h | Medical |
| 137Cs | β-, γ; 30 y | 137mBa | γ; 2.6 m | Gamma radiography & Radiation sterilization |
| 140Ba | β-, γ; 13 d | 140La | β-, γ; 40 h | Lanthanum tracer |
| 144Ce | β-, γ; 284 d | 144Pr | β-; 17 m | Calibration source |
| 210Pb | β-, γ; 21 y | 210Bi | β-; 5.0 d | Calibration source |
| 226Ra | α, 1600 y | 222Rn | α, 3.8 d | Medical |
| 238U | α, 4.5 Gy | 234Th | β-, γ; 24 d | Thorium tracer |